A nitrogen gas laser pulse with a wavelength of 337 nm contains 3.83 mJ of energy. How
Question:
A nitrogen gas laser pulse with a wavelength of 337 nm contains 3.83 mJ of energy. How many photons does it contain?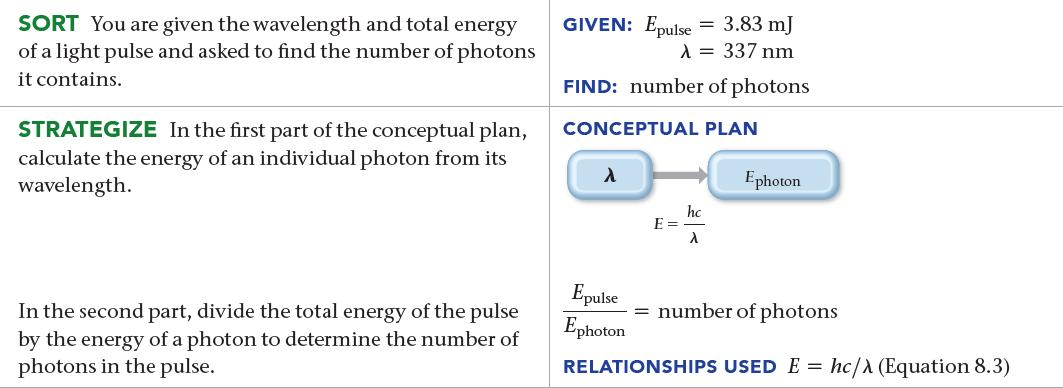
Transcribed Image Text:
SORT You are given the wavelength and total energy of a light pulse and asked to find the number of photons it contains. STRATEGIZE In the first part of the conceptual plan, calculate the energy of an individual photon from its wavelength. In the second part, divide the total energy of the pulse by the energy of a photon to determine the number of photons in the pulse. GIVEN: Epulse = 3.83 mJ λ = 337 nm FIND: number of photons CONCEPTUAL PLAN λ E = hc λ Ephoton Epulse Ephoton = number of photons RELATIONSHIPS USED E= hc/> (Equation 8.3)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
337 nm X Ephoton hc 10 m 1 nm 337 x 107 m 6626 X 1034Js 3...View the full answer

Answered By

Mishark muli
Having any assignments and any other research related work? worry less for I am ready to help you with any task. I am quality oriented and dedicated always to produce good and presentable work for the client once he/she entrusts me with their work. i guarantee also non plagiarized work and well researched work to give you straight As in all your units.Feel free to consult me for any help and you will never regret
4.70+
11+ Reviews
37+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A 1-second pulse of a red laser pointer with a wavelength of 635 nm contains 5.0 mJ of energy. How many photons does it contain?
-
A laser used to read CDs emits red light of wavelength 700 nm. How many photons does it emit each second if its power is? (a) 0.10 W, (b) LOW?
-
A laser used to weld detached retinas puts out 28-ms-long pulses of 640-nm light which average 0.68-W output during a pulse. How much energy can be deposited per pulse and how many photons does each...
-
(1) Choose all of the following statements that are correct about the time evolution of a general wave function: (I) The time evolution of a general wave function is governed by the Hamiltonian...
-
In its 2006 annual report, McDonalds Corporation reports beginning total assets of $30.0 billion; ending total assets of $29.0 billion; net sales of $21.6 billion, and net income of $3.5 billion. (a)...
-
Reflective Coatings and Herring. Herring and related fish have a brilliant silvery appearance that camouflages them while they are swimming in a sunlit ocean. The silveriness is due to platelets...
-
1. Working in small groups, complete the Pay Raise Worksheet. 2. After the class reconvenes, group spokespersons present group findings. Pay Raise Worksheet April Knepper is the new supervisor of an...
-
Selected ledger accounts for Rolm Company are given below for the just completed year: Required: 1. What was the cost of raw materials put into production during the year? 2. How much of the...
-
Based on the above case study, "Want-2-Work", present each of the following: 1) ER diagram 2) Relational model 3) Rules or assumptions made 4) Between 3 or 5 queries using the relational model...
-
Which wavelength of light has the highest frequency? a) 10 nm b) 10 mm c) 1 nm d) 1 mm
-
Which statement best describes the differences between a bright green laser and a dim red laser? (a) The two lasers emit light of the same frequency, and the light from the green laser has a greater...
-
Every month, your neighbour receives three cheques, one for 12,000, one for 7,000 and one for 3,000. The largest cheque takes 4 days to clear after it is deposited; the smallest one takes 5 days; and...
-
Discuss the impact of the Great Recession and the COVID-19 pandemic on the U.S. economy. Compare the severity of the impacts of these global economic crises. Provide examples of workplace-based human...
-
Discuss how post-pandemic shifts affect decision-making in forecasting demand and supply , planning for operations and logistics, inventory and pricing Include tools and techniques used to address...
-
Discuss how the companies that benefit most from spend analysis go beyond tactical purchasing to adopt strategic sourcing disciplines. 2.) Analyse the limitations of ERP and how Business Intelligence...
-
Discuss the difference of the Patent System between Canada and the United States. Provide recommendations that you think that the Canadian Government can do to provide greater protection and make...
-
Discuss how damage assessment may change in the future. Consider these questions in your discussion: What are the existing challenges of conducting damage assessment that would evoke a change? How...
-
Molino Inc. reported sales revenue of $2 million for 2014. Accounts receivable decreased $200,000 and accounts payable increased $300,000. Compute cash receipts from customers, assuming that the...
-
Using (1) or (2), find L(f) if f(t) if equals: t cos 4t
-
Show that two of the set of four equivalent orbitals appropriate for sp 3 hybridization, And are orthogonal. W. =-62, + 2p, + 2, + 2p,) (: - O2p. 2p, + 2p,)
-
Show that the water hybrid bonding orbitals given by a = 0.55 2Pz + 0.71 2 px -0.45 2 ps b = 0.55 2pz - 0.71 2px - 0.45 2s are orthogonal.
-
Predict which of the bent molecules, BH 2 or NH 2 , should have the larger bond angle on the basis of the Walsh correlation diagram in Figure 24.11. Explain your answer. Figure 24.11 16, 1Tu + 2a,...
-
How can Real Time Protocol (RTP) reduce cost IP network?
-
References: https://www.tbkcreative.com/ we are making a digital audit for TBK creative company please read the requirements above, pls take your time analyzing the data and name the tools used in...
-
You've learned about the product market expansion matrix. Now, it's time for you to apply what you've learned to an actual company (not a company we've discussed in class: CVS, LinkedIn, Spotify,...

Study smarter with the SolutionInn App


