Predict which of the bent molecules, BH 2 or NH 2 , should have the larger bond
Question:
Figure 24.11
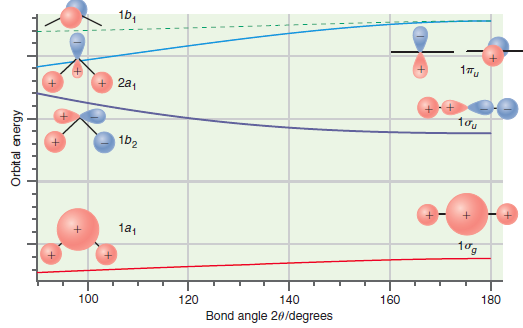
Transcribed Image Text:
16, 1Tu + 2a, 1b2 1a1 180 160 140 100 120 Bond angle 20/degrees Orbital energy
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (15 reviews)
Both molecules are equivalent through the 2a 1 1 orbital Howev...View the full answer

Answered By

Aijaz Khan
I am highly enthusiastic about tutoring. I share a friendly but professional relationship with my students. After completing my electrical engineering I actually taught a course to undergraduates for GATE exam as T.A and it was a brilliant experience and I was one among very few to finish my course on time. I have also helped professors to prepare lessons for my junior fellows while doing my undergrad. Every time I have taught so far, the response has been very heart warming. I hope to continue this and keep on improving it till I am here. Apart from this I have conducted many one on one tutoring lessons.
I believe in focussing on basic and core concepts inorder to keep students' interest alive. My only aim while tutoring is to make student understand concept in such a way that he/she can explain the learnt topic to anyone. Apart from this problem solving is my main focus while tutoring.
I hope to work with SolutionInn for long time. Hope my students will feel the difference.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict whether the ground state or the first excited state of CH 2 should have the larger bond angle on the basis of the Walsh correlation diagram shown in Figure 24.11. Explain your answer. 1b, 17u...
-
Suppose we have some optically pure (R)-2-butyl acetate that has been "labeled" with the heavy 18O isotope at one oxygen atom as shown. (a) Draw a mechanism for the hydrolysis of this compound under...
-
Predict which of the following liquids has greater surface tension: ethanol (C2H5OH) or dimethyl ether (CH3OCH3)?
-
How well employees modify their thoughts and behavior to align with and support a new or changing environment is known as Multiple Choice proactive task performance. proficient task performance....
-
Find the exact values of the remaining trigonometric functions of satisfying the given conditions. 1. tan = 15/8, sin > 0 2. cos = 8/17, tan < 0
-
Aidys Apple Pies is a roadside business. Aidy must pay $9.00 in rent each day. In addition, it costs her $1.00 to produce the first pie of the day, and each subsequent pie costs 50% more to produce...
-
When to file a motion to strike?
-
Internal Control Questionnaire Items: Assertions, Tests of Controls, and Possible Errors or Frauds. Following is a selection of items from the payroll processing internal control questionnaire in...
-
THE TONOWIlly inviation pertainS LU JAE Corporation at January 1, Itali. Common stock, $11 par, 12,000 shares authorized, 2,400 shares issued and outstanding Paid-in capital in excess of par, common...
-
Design a recursive linear-time algorithm that tests whether a binary tree satisfies the search tree order property at every node.
-
Show that the water hybrid bonding orbitals given by a = 0.55 2Pz + 0.71 2 px -0.45 2 ps b = 0.55 2pz - 0.71 2px - 0.45 2s are orthogonal.
-
Derive two additional mutually orthogonal hybrid orbitals for the lone pairs on oxygen in H 2 O, each of which is orthogonal to Ï a and Ï b , by following these steps: a. Starting with the...
-
If a taxpayer makes $30,000 per year, will the annual withholding differ depending on whether the taxpayer is paid weekly, semimonthly, or monthly? Explain.
-
(a) Using the Henderson-Hasselbalch equation, determine the amount of acetic acid (volume in mL, starting with a 1.0 M stock solution) and sodium acetate trihydrate (mass in grams) that must be mixed...
-
Sarnia Ltd . is a manufacturing company that produces a single product. The company keeps meticulous records of manufacturing activities from which the following information has been extracted: March...
-
The production department of Zan Corporation has submitted the following forecast of units to be produced by quarter for the upcoming fiscal year: 1 st Quarter 2 nd Quarter 3 rd...
-
Consider a cube with edge lengths of 1.2 inches and has a mass of 360. Calculate the density in 2m the correct number of significant numbers? 3 to C
-
Explain how a credit cardholder has an interest free loan on their new purchases. When does this occur? How can the cardholder get it ? [ Note: This has nothing to do with a promotion or enticement...
-
The following are the Year 9 income statements of Kent Corp. and Laurier Enterprises. Additional Information: Kent acquired its 40% interest in the common shares of Laurier in Year 3 at a cost of...
-
To balance the chemical equation SiH3 + O2 SiO2 + HO, you could introduce coefficients a, b, c, d and write aSiH3 + bO2 cSiO + dHO then write linear equations for each element. The equation for Si...
-
Derive an expression for the time dependence of the degree of polymerization for a stepwise polymerization in which the reaction is acid catalysed by the -COOH acid functional group. The rate law is...
-
Calculate the average polymer length in a polymer produced by a chain mechanism in which termination occurs by a disproportionation reaction of the form M + M M + :M.
-
Calculate the ratio of the mean cube molar mass to the mean square molar mass in terms of (a) The fraction p, (b) The chain length.
-
Managers use CVP concepts to perform sensitivity analysis. Sensitivity analysis is a "what-if" technique that asks what will happen to a company's breakeven or target profit if sales price, costs, or...
-
John Fleming, chief administrator for Valley View Hospital, is concerned about the costs for tests in the hospital s lab. Charges for lab tests are consistently higher at Valley View than at other...
-
A company is looking at new equipment with an installed cost of $436,859. This cost will be depreciated straight-line to zero over the project's 6-year life, at the end of which the equipment can be...

Study smarter with the SolutionInn App


