A solution contains equal concentrations of Ba 2+ , Pb 2+ , and Ca 2+ ions. When
Question:
A solution contains equal concentrations of Ba2+, Pb2+, and Ca2+ ions. When postassium sulfate is added to this solution, which cation precipitates first?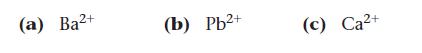
Transcribed Image Text:
(a) Ba²+ (b) Pb²+ (c) Ca²+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a Ba has the smallest solubil...View the full answer

Answered By

Ehsan Mahmood
I’ve earned Masters Degree in Business Studies and specialized in Accounts & Finance. Couple with this, I have earned BS Sociology from renowned institute of Pakistan. Moreover, I have humongous teaching experience at Graduate and Post-graduate level to Business and humanities students along with more than 7 years of teaching experience to my foreign students Online. I’m also professional writer and write for numerous academic journals pertaining to educational institutes periodically.
4.90+
248+ Reviews
287+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Generate a random variable from the p.d.f f(x)=2x:0x1 and f(x)=0:x0x1 using acceptance rejection method. Take g(x)=1,0x1 and c=2
-
A saturated solution of copper(I) sulfide, Cu 2 S, contains 1.91 10 12 g of Cu 2 S dissolved in 1 dm 3 of water. (A r values: Cu = 63.5, S = 32.1) a. Write an equilibrium expression for the...
-
Silver sulfate dissolves in water according to the reaction: A 1.5-L solution contains 6.55 g of dissolved silver sulfate. If additional solid silver sulfate is added to the solution, will it...
-
In SSL and TLS, why is there a separate Change Cipher Spec Protocol rather than including a change_cipher_spec message in the Handshake Protocol?
-
On January 2, 2013, Benson Hospital purchased a $100,000 special radiology scanner from Picard Inc. The scanner had a useful life of 4 years and was estimated to have no disposal value at the end of...
-
Describe the phase changes that take place when a liquid mixture of 4.0 mol B2H6 (melting point 131 K) and 1.0 mol CH30CH3 (melting point 135 K) is cooled from 140 K to 90 K. These substances form a...
-
Which of the following control procedures could prevent or detect errors or frauds arising from shipments made to unauthorized parties? a. Document policies and procedures for scheduling shipments....
-
At the beginning of 2016, Better Corp.s accounting records had the following general ledger accounts and balances. Better Corp. completed the following transactions during 2016: 1. Purchased land for...
-
Discuss the role of the kernel in enabling virtualization technologies. What is the difference between kernel-based virtual machines (e.g., KVM) and traditional hypervisors, and how do kernels manage...
-
Determine whether each compound is more soluble in an acidic solution than it is in a neutral solution. (a) BaF 2 (b) AgI (c) Ca(OH) 2
-
Three 15.0-mL acid samples0.10 M HA, 0.10 M HB, and 0.10 M H 2 Care all titrated with 0.100 M NaOH. If HA is a weak acid, HB is a strong acid, and H 2 C is a diprotic acid, which statement is true of...
-
Rojas Corporations comparative balance sheets are presented below. Additional information: 1. Net income was $22,630. Dividends declared and paid were $19,500. 2. No noncash investing and financing...
-
What is the function of Taskset command in Linux Ubuntu and how can you assign 2 CPU to the MySQL Ubuntu process.?
-
Changes in Current Operating Assets and Liabilities Blue Circle Corporation's comparative balance sheet for current assets and liabilities was as follows: Dec. 31, Year 2 Dec. 31, Year 1 Accounts...
-
Divide and check your result: a) 77218 by 35 b) 51362 by 245 b) 4+ 6/3 x 4-2 Simply by using DMAS: a) 12/6X4+6 Solve the following problems: c) 39-0 x 6/3 a) What should be added to 2374 to get...
-
Jean Munyutu is an African student who went to France on a trip to study to be a pastry chef. He met and became friends with Francois Dubois. After spending some time together and becoming friends,...
-
Consider the OLG model set up with two generations at every point in time. Old agents value the consumption of the current young generation, with a weight and so they consume part of their savings...
-
The contribution margin income statement of Delectable Donuts for August 2014 follows: Delectable sells four dozen plain donuts for every dozen custard-filled donuts. A dozen plain donuts sells for $...
-
The following selected accounts and normal balances existed at year-end. Notice that expenses exceed revenue in this period. Make the four journal entries required to close the books: Accounts...
-
Sketch Bode magnitude and phase plots for 1.6 s = jo H(s) = s(s + s + 16)
-
Construct the Bode magnitude and phase plots for 2(s + 1) H(s) = s = ja (s + 2)(s + 10)*
-
Draw the Bode plots for 250(jo + 1) jo(-a + 10 ja + 25) ) -
-
Suppose we are comparing two cities, A and B. Cities A and B are the same in every respect, other than city A having a larger employment multiplier than city B. Using two graphs, one for city A and...
-
What is the final equilibrium temperature when 17 g of milk at 11C is added to 100 g of coffee at 61C? Assume the specific heats of milk and coffee are the same as that of water, and neglect the...
-
Cite relevant arguments found in the Supreme Court ruling and your interpretation of the data to support your answers to the following questions in the form of a memo: 1) How does the provided data...

Study smarter with the SolutionInn App


