An aspirin tablet contains 325 mg of acetylsalicylic acid (C 9 H 8 O 4 ). How
Question:
An aspirin tablet contains 325 mg of acetylsalicylic acid (C9H8O4). How many acetylsalicylic acid molecules does it contain?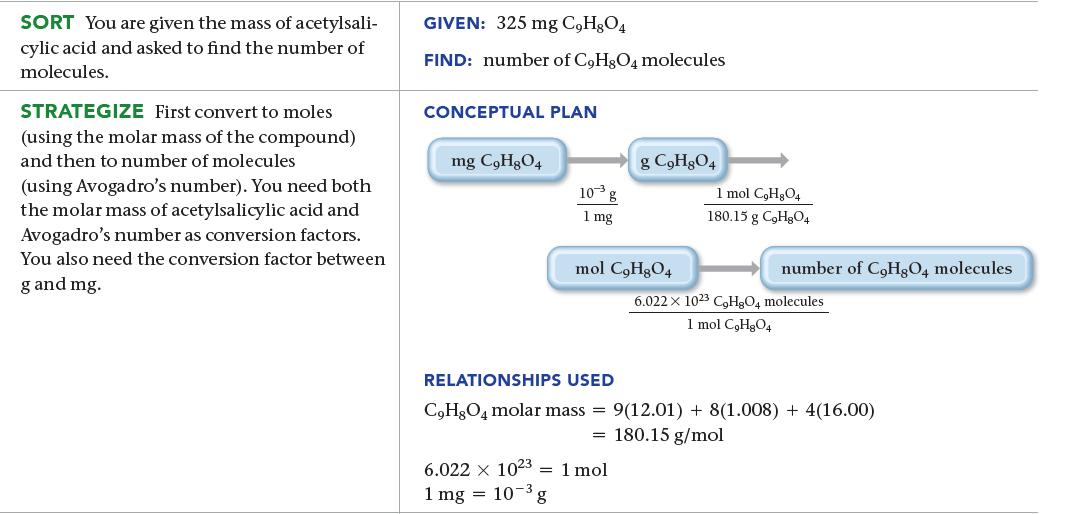
Transcribed Image Text:
SORT You are given the mass of acetylsali- cylic acid and asked to find the number of molecules. STRATEGIZE First convert to moles (using the molar mass of the compound) and then to number of molecules (using Avogadro's number). You need both the molar mass of acetylsalicylic acid and Avogadro's number as conversion factors. You also need the conversion factor between g and mg. GIVEN: 325 mg C,H,O4 FIND: number of C9H8O4 molecules CONCEPTUAL PLAN mg C₂H₂O4 10³ g 1 mg g C₂H8O4 mol C₂H₂O4 6.022 x 1023 1 mol 1 mg = 10-3, ³g 1 mol C₂H₂O4 180.15 g C₂H8O4 number of CoHgQ4 molecules 6.022 x 1023 C₂H8O4 molecules 1 mol C₂H₂O4 RELATIONSHIPS USED C₂H8O4 molar mass= 9(12.01) + 8(1.008) + 4(16.00) = 180.15 g/mol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
325 mg CH8O4 X 10 g 1 m...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A typical aspirin tablet contains 325 mg acetylsalicylic acid (HC 9 H 7 O 4 ). Calculate the pH of a solution that is prepared by dissolving two aspirin tablets in enough water to make one cup (237...
-
Given f(x) is a polynomial function with leading term of -24x^(11), a. How many zeros will f(x) have? b. How many x-intercept s can f(x) have? c. How many turning points will f(x) have?
-
Given a rigid tank with V=800 L filled with m = 2 kg of HO at P = 0.2 MPa. Write your answers in the table below. Only answers in the table are counted. Specific volume (m/kg) - R Phase description:...
-
What is marginal cost Explain with an example.
-
In the previous problem, suppose your required return on the project is 20 percent and your pretax cost savings are $340,000 per year. Will you accept the project? What if the pretax cost savings are...
-
Mama Leones Frozen Pizzas uses 50,000 units of cheese per year. Each unit costs $2.50. The ordering cost for the cheese is $250 per order, and its carrying cost is $0.50 per unit per year. Calculate...
-
Use the Hawkeye Gymnastics data in Exercise 16-22 to compute a. New borrowing or payment of long-term notes payable, with Hawkeye having only one long-term note payable transaction during the year b....
-
A firm wishes to minimize annual inventory costs. The firm uses the EOQ model to determine the cost-minimizing order quantity and the reorder point. Annual demand, units 22,100 Item cost, $ per unit...
-
You are pulling a box across a rough surface. What are the forces on the box? What are the forces on you? Draw a free body diagram of each object. Identify action-reaction pairs. Why do...
-
Google is considering a project to move a fraction of its workforce out of Silicon Valley to a cheaper, more efficient location. There are two alternatives. Project 1: the initial move costs $45...
-
What is the formula mass for a compound? Why is it useful?
-
A compound has the empirical formula CH 2 O and a formula mass of 120.10 amu. What is the molecular formula of the compound? a) CH 2 O b) C 2 H 4 O 2 c) C 3 H 6 O 3 d) C 4 H 8 O 4
-
Perfect Furniture is a manufacturer of kitchen tables and chairs. The company is currently deciding between two new methods for making kitchen tables. The first process is estimated to have a fixed...
-
Delineate the differences between components, entities, items, and CIs and their Entity Relationships (ERs)?
-
Contrast judgmental and statistical sampling.
-
What do you think led mankind to embark upon the study of Artificial Intelligence? Which fields of study particularly fed into it? What human desires did the study of Artificial Intelligence seek to...
-
What problems with existing Engineering paradigms is it intended to correct?
-
A PV battery system has an end-to-end efficiency of \(77 \%\). The system is used to run an all-AC load that is run only at night. The charge controller efficiency is \(96 \%\) and the inverter...
-
Consider the following sales forecasts for products A and B: Each unit of product A takes approximately 2.5 labor hours, while each unit of product B takes only 1.8 hours. a. What is the combined...
-
Kenneth Hubbard has prepared the following list of statements about managerial accounting and financial accounting. 1. Financial accounting focuses on providing information to internal users. 2....
-
For 1.25 mol of an ideal gas, P external = P =350. 10 3 Pa.The temperature is changed from 135C to 21.2C, and C V ,m = 3/2R. Calculate q, w, U, and H.
-
Suppose an adult is encased in a thermally insulating barrier so that all the heat evolved by metabolism of foodstuffs is retained by the body. What is her temperature increase after 2.5 hours?...
-
Draw bond-line structures for all constitutional isomers of C 5 H 12 ?
-
Go into depth about the topic: Do women in the workplace still have to tolerate sexual harassment to advance or maintain their job positions? Why or why not? THE SCENARIO: You are human resource...
-
A pile group consists of nine friction piles in clay soil (see Figure 10-40). The diameter of each pile is 16 in., and the embedded length is 30 ft each. Center-to-center pile spacing is 4 ft. Soil...
-
Honest to Goodness Merchandising was registered as a Single Proprietorship by May Mendoza, the owner in the Department of Trade and Industry. Selected transactions for the month of July, 2019 are...

Study smarter with the SolutionInn App


