Balance each redox reaction occurring in acidic aqueous solution. a. I (aq) + NO (aq) b. CIO4
Question:
Balance each redox reaction occurring in acidic aqueous solution.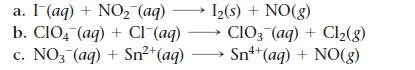
Transcribed Image Text:
a. I (aq) + NO₂ (aq) b. CIO4 (aq) + Cl¯(aq) c. NO3(aq) + Sn²+ (aq) 1₂(s) + NO(g) CIO3(aq) + Cl₂(g) Sn+ (aq) + NO(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
a Iaq NOaq Is NOg Halfreactions Oxidation Iaq Is 2e Reduction NOaq Haq e NOg HOl Balance the oxidati...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
PART A: Balance each redox reaction occurring in acidic aqueous solution. MnO4( a q )+Al( s )Mn2+( a q )+Al3+( a q ). Express your answer as a chemical equation. Identify all of the phases in your...
-
Balance each redox reaction occurring in acidic aqueous solution. a. PbO(s) + I (aq) Pb+ (aq) + 1(s) 2+ b. SO32 (aq) + MnO4 (aq) SO42 (aq) + Mn+ (aq) 2- c. S03 (aq) + Cl(g) SO4 (aq) + Cl (aq)
-
Balance each redox reaction occurring in acidic aqueous solution. a. Zn(s) + Sn+ (aq) b. Mg(s) + Cr+ (aq) Zn+ (aq) + Sn(s) Mg+ (aq) + Cr(s) c. MnO4 (aq) + Al(s) Mn+ (aq) + A1+ (aq)
-
You require inventory and accounts receivable collateral for all C&I loans. You have a guideline of an advance rate of 70% for customer receivables of less than 60 days of age. Older receivables get...
-
Lago Corporation is considering adopting the standard costing method. Dan Sarkis, the manager of the Ohio Division, attended a corporate meeting at which Leah Rohr, the controller, discussed the...
-
Can you describe a revenue recognition setting in financial accounting that employs the concept of equivalent work?
-
Inland Equipment uses the allowance method to account for uncollectible accounts. On December 31,2010, Allowance for Uncollectible Accounts has a $600 credit balance. Journalize the year-end...
-
Alpha Company is considering the purchase of Beta Company. Alpha has collected the following data about Beta: Cumulative total net cash earnings for the past five years of $850,000 includes...
-
Describe the components of a system changer's leadership style, evaluating how working in a global economy may impact the function of the organization.
-
Balance each redox reaction occurring in basic aqueous solution. a. HO(aq) + ClO(aq) CIO (aq) + O(g) b. Al(s) + MnO4 (aq) MnO(s) + Al(OH)4 (aq) c. Cl(g) Cl(aq) + CIO (aq)
-
How can the corrosion of iron be prevented?
-
Having just graduated with a business degree, you're excited to begin working as a junior accountant at Sunny Optics, Inc. The company supplies lenses, frames, and sunglasses to opticians and...
-
Evaluate 49 Enter DNE if the result is not a real number.
-
For each of the resource types mentioned below, give an example of a relevant Vic legislation and policy or procedure. Explain how the absence of the legislation and policy/procedure would have an...
-
How do we account for our human assets in our accounting of the company? What typically shows up regarding this matter?
-
Provide 2 paragraphs what are the reasons for Apple's successes? with text citations and accessible references based on APA style.
-
Nike's revenue for the 12 months ending May 31, 2022, which increased by 9.5% from the prior year, was $46.71 billion . Strong demand for Nike products in North America and China was the primary...
-
A condensed income statement for Southwest Airlines and a partially completed vertical analysis are presented below. Required: 1. Complete the vertical analysis by computing each line item (a)-(f) as...
-
Keating & Partners is a law firm specializing in labour relations and employee-related work. It employs 25 professionals (5 partners and 20 managers) who work directly with its clients. The average...
-
Estimate the pressure exerted by your feet when you stand upright on the floor. Compare it to the pressure when you are wearing stiletto heels (use the value at the heel).
-
When traveling in a commercial airplane, you are sometimes given snacks, such as peanuts, in a sealed bag. The bag is often bulging much more than when you purchase a bag of peanuts in a store....
-
When a typical 1200-page textbook is sitting on a table, it exerts a force on the table. Estimate the associated pressure.
-
The Atlanta Food Bank is a nonprofit organization that receives donations of food and distributes this food to appropriate charitable organizations. View the additional information. He used this wait...
-
Check my work As the chief financial officer of Adirondack Designs, you have the following information: Next year's expected net income after tax but before new financing Sinking-fund payments due...
-
Huegel Hollow Resort has ordered 24 rotomolded kayaks from Current Designs. Each kayak will be formed in the rotomolded oven, cooled, and then have the excess plastic trimmed away. Then, the hatches,...

Study smarter with the SolutionInn App


