Balance each redox reaction occurring in acidic aqueous solution. a. Zn(s) + Sn+ (aq) b. Mg(s) +
Question:
Balance each redox reaction occurring in acidic aqueous solution.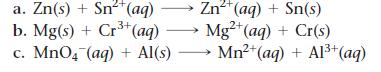
Transcribed Image Text:
a. Zn(s) + Sn²+ (aq) b. Mg(s) + Cr³+ (aq) — Zn²+ (aq) + Sn(s) Mg²+ (aq) + Cr(s) c. MnO4 (aq) + Al(s) →→→ Mn²+ (aq) + A1³+ (aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
To balance redox reactions occurring in acidic aqueous solution you can follow these steps Identify the oxidation and reduction halfreactions Balance ...View the full answer

Answered By

Aun Ali
I am an Associate Member of Cost and Management Accountants of Pakistan with vast experience in the field of accounting and finance, including more than 17 years of teaching experience at university level. I have been teaching at both undergraduate and post graduate levels. My area of specialization is cost and management accounting but I have taught various subjects related to accounting and finance.
5.00+
13+ Reviews
32+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
PART A: Balance each redox reaction occurring in acidic aqueous solution. MnO4( a q )+Al( s )Mn2+( a q )+Al3+( a q ). Express your answer as a chemical equation. Identify all of the phases in your...
-
Balance each redox reaction occurring in acidic aqueous solution. a. I (aq) + NO (aq) b. CIO4 (aq) + Cl(aq) c. NO3(aq) + Sn+ (aq) 1(s) + NO(g) CIO3(aq) + Cl(g) Sn+ (aq) + NO(g)
-
Balance each redox reaction occurring in acidic aqueous solution. a. PbO(s) + I (aq) Pb+ (aq) + 1(s) 2+ b. SO32 (aq) + MnO4 (aq) SO42 (aq) + Mn+ (aq) 2- c. S03 (aq) + Cl(g) SO4 (aq) + Cl (aq)
-
Assume that n is a positive integer. For each of the following algorithm segments, how many times will the innermost loop be iterated when the algorithm segment is implemented and run? 1) for k:=1 to...
-
Using the data in E 10 and the average costing method, prepare a process cost report that computes the equivalent units for November, the product unit cost for the toys, and the ending balance in the...
-
Sriram Motor Works manufactures scooters and other two-wheelers for sale in Asia. The firm has divided its operations into two main factories: one in Pune, India, and the other in Malaysia. The firm...
-
Rice Automotive ended December 2009 with Accounts Receivable of $30,000 and Allowance for Uncollectible Accounts of $1,500. During January 2010, Rice Automotive completed the following transactions:...
-
Aretail store in Des Moines, Iowa, receives shipments of a particular product from Kansas City and Minneapolis. Let x = units of product received from Kansas City y = units of product received from...
-
For XYZ Printing Company, the following costs have been identified Customer Complaint remakes $27,000 Printing Plate revisions $28,000 Quality improvement projects $13,000 Gauging $100,000 Other...
-
Balance each redox reaction occurring in basic aqueous solution. a. HO(aq) + ClO(aq) CIO (aq) + O(g) b. Al(s) + MnO4 (aq) MnO(s) + Al(OH)4 (aq) c. Cl(g) Cl(aq) + CIO (aq)
-
How can the corrosion of iron be prevented?
-
Buster Bluelip, a student repeating organic chemistry for the fifth time, has observed that alcohols can be converted into alkyl bromides by treatment with concentrated HBr. He has proposed that, by...
-
1. Transportation is a derived demand; discuss, citing its role in economic advancement and stability.? 2. Transportation enables mobility globally however it creates daunting and damaging...
-
Please give me a very brief summary of a segment of the NYC rental market over the last five years. For example what has happened to the prices and availability of rental units in Greenpoint...
-
Look at the balance sheet for the last two years. What caused the changes in the balance sheet from last year to this year?Use account names and numbers for both years to explain your answer. Look at...
-
Often times, concluding on the severity of a deficiency requires a conversation with the client. What are some questions that you might ask the client in order to assess the severity of any potential...
-
Answer the following thouroughly and put websites or references: What are the key features of a computerised accounting system? a) What proof/substantiation documents are available to support...
-
Use the data given in PB13-1 for Tiger Audio. Required: 1. Compute the gross profit percentage in 2013 and 2012. Is the trend going in the right direction? 2. Compute the net profit margin for 2013...
-
"Standard-cost procedures are particularly applicable to process-costing situations." Do you agree? Why?
-
Express a pressure of 5.0 10 6 Pa in units of pounds per square inch.
-
An altimeter is a device used to measure altitude. Most altimeters are based on measurements of air pressure. Explain how they work and estimate how much the pressure changes for a 100-m change in...
-
The water in the pipes of a typical house is at a gauge pressure of 3.0 times atmospheric pressure. What is the absolute pressure in pascals and in units of pounds per square inch?
-
Compute the missing amount in the accounting equation for each entity from the financial information presented: Assets Liabilities Equity Your Basket $ ? $ 28,000 46,000 Flowers and Gifts 85,000 ?...
-
9. Identify the CIA area(s) affected in each of these incidents and some actions organizations can take to ensure that these incidents do not happen to them. Incident 414's: Confidentiality. Morris...
-
Fuel taxes (paid by filling up a vehicle with gasoline or diesel) accounts for approximately 70% of the funds that are used to build and maintain roads. As the number of electric vehicles continues...

Study smarter with the SolutionInn App


