Based on their molecular structure, pick the stronger acid from each pair of binary acids. Explain your
Question:
Based on their molecular structure, pick the stronger acid from each pair of binary acids. Explain your choice.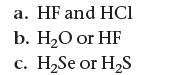
Transcribed Image Text:
a. HF and HCI b. H₂O or HF c. H₂Se or H₂S
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a HCl weake...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Based on their molecular structure, pick the stronger acid from each pair of oxyacids. Explain your choice. a. HSO4 or HSO3 c. HCIO or HBrO b. HClO or HCIO d. CC13COOH or CH3COOH
-
Based on their molecular structure, pick the stronger acid from each pair of oxyacids. Explain your choice. MISSED THIS? Read Section 17.10 a. HSO4 or HSO3 c. HCIO or HBrO b. HClO or HCIO d. CCI,COOH...
-
Sam's Insurance must choose between two types of printers. Both printers meet the firm's quality standard. Printer A costs $3,500 and is expected to last 3 years with operating costs of $380 per...
-
Acceleration is the rate of change of velocity with time. Is the acceleration vector always aligned with the velocity vector? Explain.
-
Golding Company produces leather strips for western belts using three processes: cutting, design and coloring, and punching. The weighted average method is used for all three departments. The...
-
(a) Express the vector field in cylindrical and spherical coordinates, H = xy2zax + x2yzay + xyz2az (b) In both cylindrical and spherical coordinates, determine H at (3, 4, 5).
-
The Cooper Furniture Company of Potomac, Maryland, assembles two types of chairs (Recliners and Rockers). Separate assembly lines are used for each type of chair. Classify each cost item (AI) as...
-
C-Cell Wireless needed additional capital to expand, so the business incorporated. The charter from the state of Georgia authorizes C-Cell to issue 50,000 shares of 7%, $50 par value cumulative...
-
True regarding amendments that propose to change the proportional percentage by which each condominium or cooperative member shares common areas expenses or owns the common surplus in Florida?
-
Based on molecular structure, arrange the binary compounds in order of increasing acid strength. Explain your choice. HTe, HI, HS, NaH
-
Calculate the [H 3 O + ] and pH of each H 2 SO 4 solution. At approximately what concentration does the x is small approximation break down? a. 0.50 M b. 0.10 M c. 0.050 M
-
A college has decided to introduce the use of plus and minus with letter grades, as long as there is convincing evidence that more than 60% of the faculty favor the change. A random sample of faculty...
-
What are demand shocks? Distinguish between positive and negative demand shocks.
-
What causes the long-run aggregate supply curve to shift?
-
Describe the adjustment to long-run equilibrium if an economys short-run equilibrium output is above potential output.
-
How does the condition for short-run equilibrium differ from that for long-run equilibrium?
-
Describe the five time lags involved in implementing stabilization policy.
-
Adria Lopez created Success Systems on October 1, 2013. The company has been successful, and Adria plans to expand her business. She believes that an additional $ 86,000 is needed and is...
-
Figure displays a 12.0 V battery 3 four uncharged capacitors of capacitances C1 = 4.00F, C2 = 6.00F, and C3 = 3.00F. The switch is thrown to the left side until capacitor 1 is fully charged. Then the...
-
A compound lens is composed of two thin lenses separated by 10 cm. The first of these has a focal length of +20 cm, and the second a focal length of -20 cm. Determine the focal length of the...
-
A convex-planar lens of index 3/2 has a thickness of 1.2 cm and a radius of curvature of 2.5 cm. Determine the system matrix when light is incident on the curved surface.
-
A thick biconvex lens in air has an index of 1.810 and a thickness of 3.00 cm. Its first radius of curvature is 11.0 cm and its second is 120 cm. Determine its system matrix A.
-
Additional financial statement information $ millions 2018 2017 Average equity $2,929.0 $2,347.0 a. Compute the return on equity for each year. Note: 1. Select the appropriate numerator and...
-
Sarah: While working towards her graduate degree in the applied sciences, Sarah is work- ing for a small firm that requires their staff, who work out of the company office, to invoice the company for...
-
Raner, Harris and Chan is a consulting firm specializing in information systems for medical and dental clinics. The firm has two offices-one in Chicago and one in Minneapolis. It classifies the...

Study smarter with the SolutionInn App


