Calculate E cell for each balanced redox reaction and determine if the reaction is spontaneous as written.
Question:
Calculate E°cell for each balanced redox reaction and determine if the reaction is spontaneous as written.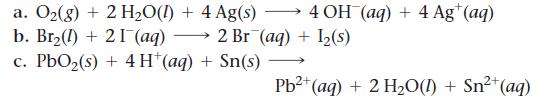
Transcribed Image Text:
a. O₂(g) + 2 H₂O(1) + 4 Ag(s) b. Br₂(1) 21 (aq) c. PbO₂ (s) + 4H+ (aq) + Sn(s) 4 OH(aq) + 4 Ag+ (aq) 2 Br (aq) + 1₂(s) 2+ Pb²+ (aq) + 2 H₂O(1) + Sn²+ (aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Reaction Ecell V Spontaneous O2g 2 H2Ol 4 Ags 4 OHaq 4 Agaq 0000 ...View the full answer

Answered By

John Kimutai
I seek to use my competencies gained through on the job experience and skills learned in training to carry out tasks to the satisfaction of users. I have a keen interest in always delivering excellent work
4.70+
11+ Reviews
24+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Calculate E cell for each balanced redox reaction and determine if the reaction is spontaneous as written. 2+ a. 2 Cu(s) + Mn+ (aq) b. MnO(aq) + 4H+ (aq) + Zn(s) c. Cl(g) + 2 F (aq) 2 Cut (aq) +...
-
Toplob Inc. ("Toplob") provides employment consulting services. These services range from maintaining payroll records to taxation services, as well as general business advisory and consulting to new...
-
What is the importance of developing plans to achieve organizational goals? explain with examples
-
Design Architects, LLP, billed clients for 6,000 hours of design work for the month. Actual variable overhead costs for the month were $315,000, and 6,250 hours were worked. At the beginning of the...
-
What are multinational corporations?
-
The plaintiff, Thelma Agnes Smith, lived with the defendant out of wedlock for several years. When the relationship ended, she sued the defendant, seeking to enforce two written agreements with him...
-
Wempe Co. sold $3,000,000, 8%, 10-year bonds on January 1, 2014. The bonds were dated January 1, 2014, and pay interest on January 1. The company uses straight-line amortization on bond premiums and...
-
The bar BC exerts a force on the cables at B that is directed along the line from C to B. (Figure 1) The mass of the suspended load is 190 kg. Suppose that = 20 and = 20. Part A What is the tension...
-
Which metal cation is the best oxidizing agent? a. Pb+ b. Cr+ c. Fe+ d. Sn+
-
Determine whether or not each metal dissolves in 1 M HIO 3 . For those metals that do dissolve, write a balanced redox equation for the reaction that occurs. a. Au b. Cr
-
Pitchford Company adopted a defined benefit pension plan on January 1, 2019, at which time it awarded retroactive benefits to its employees. The following information is available in regard to this...
-
Waystar Corporation went public last year. The IPO offered 4,000,000 shares for sale at $2.30 per share and was fully subscribed. On the first day of trading the stock price rose to $7.11. How much...
-
, 7 years ago, the City of Calabasas issued $5,000,000 of 10.5% coupon, 20-year annual payment, tax-exempt muni bonds. The bonds had 5 years of call protection, but now the bonds can be called if the...
-
Broward Manufacturing recently reported the following information: Net income $765,000 ROA 7% Interest expense $244,800 Accounts payable and accruals $950,000 Broward's tax rate is 30%. Broward...
-
Corporation's stock had a required return of 11.50% last year, when the risk-free rate was 5.50% and the market risk premium was 4.75%. Now suppose there is a shift in investor risk aversion, and the...
-
Describe a potential application scenario in which the ANSI-SPARC three-level architecture takes effect, and explain how or why.
-
Define discovery, and identify and explain five of the most important forms of discovery.
-
Why are stocks usually more risky than bonds?
-
Figure P.11.25 depicts a single saw tooth function and its convolution. Note that the convolution is asymmetricalexplain why thats reasonable. Why does the convolution begin at 0? How wide is the...
-
Graphically convolve the two functions Æ(x) and h(x) shown in Fig. P.11.26. Fig. P.11.26. How wide will the convolution be? Will it be symmetrical? Where will it start? h(x) f(x) 1 +1 -2 +2 -1
-
Graphically convolve, at least approximately, the two functions shown in Fig. P.11.31. Does that solution remind you of anything? Why is the convolution symmetrical? When does its peak value occur in...
-
Overview A company is operating in the retailing environment that sells hardware (e.g., Home Depot or Lowe's). As a marketing manager, it is one's responsibility for assessing potential risks in a...
-
a) Economic Order Quantity (EOQ) is the order quantity that minimises the sum of both the holding and ordering costs under the fixed-order quantity system. The formula of the Economic Order Quantity...
-
Sueos en Teca is a small local manufacturer of wooden benches. Their line includes four types of benches that differ in size, material, finish, and color. Pertinent production data are as follows:...

Study smarter with the SolutionInn App


