Calculate the solubility of Zn(OH) 2 (s) in 2.0 M NaOH solution. You must take into account
Question:
Calculate the solubility of Zn(OH)2(s) in 2.0 M NaOH solution.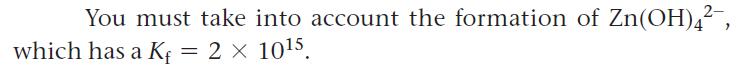
Transcribed Image Text:
You must take into account the formation of Zn(OH)4², 2 x 10¹5. which has a Kf =
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
To calculate the solubility of ZnOH2s in 20 M NaOH solutionwe must take into account the formation o...View the full answer

Answered By

ABHISHEK GAUTAM
I have an experience for teaching of two years and also i was working on chegg india for last 6 months as SME(subject matter expert) in mechanical engg. subject.I have a great passion for study and teaching .I want to make my career in teaching line itself.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following income statements illustrate different cost structures for two competing companies: Number of customers (a) Sales revenue (a x $230) Variable cost (ax $180) Variable cost (a x $0)...
-
A rigid bar ABCD is being supported by a hinge at A and two cable CE and DE. A measuring gauge determined that the vertical deflection at the free end of the bar is [y] mm. Consider these values: H =...
-
Harvatin Group reported net income totaling $1,000,000 for the year 2006. The following is additional information obtained from the Harvatin Group's financial reports: The Company purchased 100,000...
-
The two roots of a quadratic equation ax 2 + bx + c = 0 can be obtained using the following formula: b 2 - 4ac is called the discriminant of the quadratic equation. If it is positive, the equation...
-
Using 2008 as the base year, prepare a trend analysis of the following data, and tell whether the situation shown by trends is favorable or unfavorable. 2012 20 2010 2009 2008 Net sales Cost of goods...
-
Jerry is the project manager for a team developing a retail store management system for a chain of sporting goods stores. Company headquarters is in Las Vegas, and the chain has 27 locations...
-
Use the American Reserve Rare Coins data from Problem 16-33A. Requirements 1. Prepare American Reserve Rare Coins' income statement for the year ended December 31, 2006. Use the single-step format,...
-
Refer to Exercise 5-4 and prepare the appropriate journal entries for Baker Co. to record the May 5 purchase and each of the three separate transactions a through c. Baker is a retailer that uses a...
-
You work for Frederick and Sons Company (FSC) as the VP Finance. FSC's CEO (your boss), I.M. Confewzed, is currently sitting in your office with the financial statements that you prepared for 2017...
-
Explain why [Ni(NH 3 ) 4 ] 2+ is paramagnetic, while [Ni(CN) 4 ] 2- is diamagnetic.
-
Sulfide (S 2- ) salts are notoriously insoluble in aqueous solution. a. Calculate the molar solubility of nickel(II) sulfide in water. Ksp(NiS) = 3 x 10-16 b. Nickel(II) ions form a complex ion in...
-
Joseph Ohara opens a dental practice. During the first month of operation (March), the practice, titled Joseph Ohara Dental Clinic Ltd., experienced the following events: {Requirement} Dr. Ohara has...
-
The preparation of styrene (C8H8) and benzene (C6H6) from acetylene (C2H2 ) have been considered by Tanaka et al. (1955). and for this system, a visual representation of the atomic matrix is given by...
-
The manager of a carpet store is trying to determine the best installation crew size. He has tried various crew sizes with the results shown below. Calculate the average productivity when he uses a...
-
Solve If you invest $12,000 today, how much will you have in the time and at the rate indicated below? NOTE: Enter amounts rounded to two decimals (e.g., 78.76 or 40.00). PV $12,000 Rate 9% Time FV...
-
After reading the Journal attached below, studentsshould write an academic essay of about 500(+/- 10%) words on "A Critical Analysis of the impacts ofdifferent economic and political systems...
-
Primare Corporation provided the following data for last month s manufacturing operations. Purchases of raw materials $ 3 0 , 0 0 0 Indirect materials used in production $ 5 , 0 0 0 Direct labor $ 5...
-
Samantha is the production manager for Wentworth Company. Each year, she is involved in the companys budgeting process. Company President Leslie has asked Samantha to submit the facilitys budgeted...
-
What is the shape of the exponential distribution?
-
In a popular demonstration of induced emf, a lightbulb is connected across a large inductor in an RL circuit, as shown in Fig. 27.37. When the switch is opened, the bulb flashes brightly and may even...
-
You have a fixed length of wire to wind into an inductor. Will you get more inductance if you wind a short coil with large diameter, or a long coil with small diameter?
-
A car battery has a 12-V emf, yet energy from the battery provides the 30,000-V spark that ignites the gasoline. How is this possible?
-
According to the case of leonard's Carrying CO v Asiatic Pertroleum [1915] AC 705, Why is a corporation only an abstract idea and who actually comprises the directing mind and will of a corporation?
-
Discuss the following topics in relation to carriers and modulation - remember to supply a diagram to facilitate your discussion! The electromagnetic spectrum Amplitude modulation Frequency...
-
1. Consider GF(24) defined via the irreducible polynomial f(x)=X+X3+1 (note that this is not the "standard" degree-4 polynomial). (a) Write a for i = 1,..., 15 as a polynomial in a of degree at most...

Study smarter with the SolutionInn App


