Calculate the vapor pressure at 25 C of a solution containing 99.5 g sucrose (C 12 H
Question:
Calculate the vapor pressure at 25 °C of a solution containing 99.5 g sucrose (C12H22O11) and 300.0 mL water.
The vapor pressure of pure water at 25 °C is 23.8 torr. Assume the density of water is 1.00 g/mL.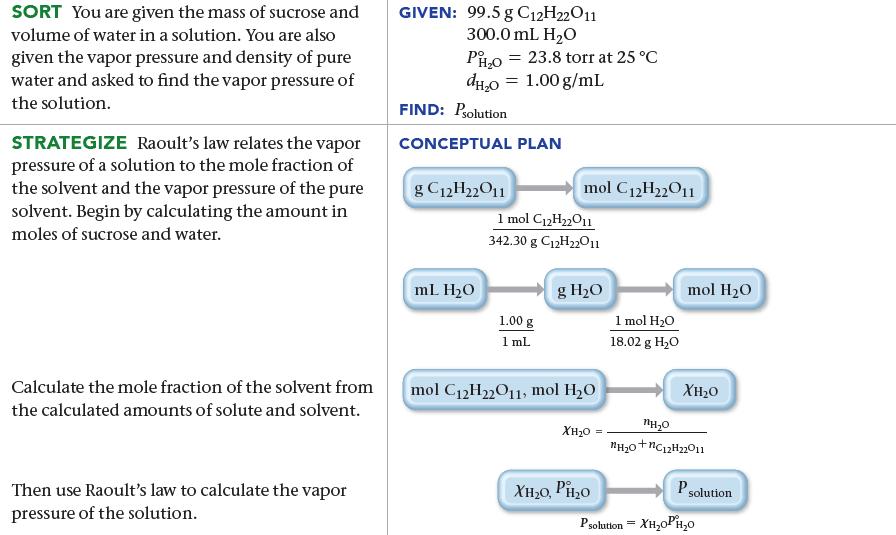
Transcribed Image Text:
SORT You are given the mass of sucrose and volume of water in a solution. You are also given the vapor pressure and density of pure water and asked to find the vapor pressure of the solution. STRATEGIZE Raoult's law relates the vapor pressure of a solution to the mole fraction of the solvent and the vapor pressure of the pure solvent. Begin by calculating the amount in moles of sucrose and water. Calculate the mole fraction of the solvent from the calculated amounts of solute and solvent. Then use Raoult's law to calculate the vapor pressure of the solution. GIVEN: 99.5 g C12H22O11 300.0 mL H₂O PH₂O dH₂0 FIND: Psolution CONCEPTUAL PLAN g C12H22011 mL H₂O = 23.8 torr at 25 °C 1.00 g/mL mol C12H22011 1 mol C₁2H₂2011 342.30 g C12H22011 1.00 g 1 mL g H₂O mol C12H22011, mol H₂O XH₂0 XH,O, PH,o 1 mol H₂O 18.02 g H₂O mol H₂O XH₂O PHO *Hạo+nC_zHzzO11 P solution Psolution = XH₂OPH₂0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
995 g C12H22O11 X 3000 mL HO x XHO nHO nC12H2...View the full answer

Answered By

Allan Simiyu
I am an adroit Writer. I am a dedicated writer having worked as a writer for 3 years now. With this, I am sure to ace in the field by helping students break down abstract concepts into simpler ideas.
5.00+
8+ Reviews
54+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Calculate the vapor pressure at 25 C of a solution containing 165 g of the nonvolatile solute, glucose, C 6 H 12 O 6 , in 685 g H 2 O. The vapor pressure of water at 25 C is 23.8 mmHg.
-
Determine the vapor pressure at 25 C of an aqueous ethylene glycol (C 2 H 6 O 2 ) solution that is 14.8 % C 2 H 6 O 2 by mass. The vapor pressure of pure water at 25 C is 23.8 torr. a) 3.52 torr b)...
-
Calculate the vapor pressure at 35C of a solution made by dissolving 20.2 g of sucrose, C12H22O11, in 70.1 g of water. The vapor pressure of pure water at 35C is 42.2 mmHg. What is the vapor-pressure...
-
Where is the line in the sand the point where such behaviors are so destructive that you feel that the relationship needs to end?
-
Life-cycle cost reduction is best achieved during the development stage of the production life cycle. Do you agree or disagree? Explain.
-
A chain with uniform mass density per unit length hangs between two supports located at the same height, a distance 2d apart (see Fig.). What should the length of the chain be so that the magnitude...
-
Im gathering some information about the sales/collection process and how it is supposed to work. Okay?
-
Here are comparative statement data for Prince Company and King Company, two competitors. All balance sheet data are as of December 31, 2014, and December 31, 2013. Instructions (a) Prepare a...
-
Your web application's database is MySQL.How you can establish the database connection and if database connection is failed, you able to detect the errors that cause the database connection failed....
-
A sodium nitrate solution is 12.5% NaNO 3 by mass and has a density of 1.02 g/mL. Calculate the molarity of the solution. a) 1.44 M b) 12.8 M c) 6.67 M d) 1.50 M
-
A solution is saturated in both nitrogen gas and potassium bromide at 75 C. When the solution is cooled to room temperature, what is most likely to happen? (a) Some nitrogen gas bubbles out of...
-
Consider the clay sample in Problem 12.10. A consolidated-undrained triaxial test was conducted on the same clay with a chamber pressure of 103 k N/m2. The pore pressure at failure (ud)f = 33 kN/m2....
-
Monique has just returned from her first business trip to Japan. While there, she was impressed with the intense use of work teams. Now back in Quebec, she would like to totally reorganize the...
-
LB Corporation purchased a business car in June 2019 for $62,000. The car weighs 4,000 pounds and will be used 100 percent of the time in the business. Compute the largest depreciation deduction...
-
What is active participation?
-
In March, Gary Parker started a new career as a real estate agent. He established Gary Parker Company, LLC on March 11 and began seeking clients. He listed his first property on March 21, and closed...
-
How can nonrental undertakings be combined?
-
Best Ink produces ink- jet printers for personal computers. It received an order for 500 printers from a customer. The following information is available for this order. Process time . . . . . . . ....
-
Explain what is meant by vicarious liability and when it is available?
-
What pressure is required above the water in Fig. 6.13 to cause the jet to rise to 9.50 m? The water depth is 1.50 m. 40.0 ft Air pressure h = 6.0 ft
-
What pressure is required above the water in Fig. 6.12 to cause the jet to rise to 28.0 ft? The water depth is 4.50 ft. h
-
To what height will the jet of water rise for the conditions shown in Fig. 6.40? p= 12.0 psig Jet 3.50 ft 3 in 9 in
-
Problem 2. Given: = (xi) is a scalar field and A, is a constant, symmetric, second- order tensor such that 6 = 1 xi xj Aij. a. Express eq. (1) in matrix notation. b. Calculate Ok and express the...
-
3. The volume of this package is 150 cm. 6 cm Granola Bits 5 cm Determine the length of the package.
-
1 Find the value of x if log 1 = x. 16 4

Study smarter with the SolutionInn App


