Complete and balance each acidbase equation. a. HI(aq) + LiOH(aq) b. HCHO(aq) + Ca(OH)(aq) c. HCl (aq)
Question:
Complete and balance each acid–base equation.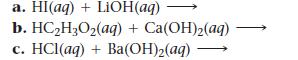
Transcribed Image Text:
a. HI(aq) + LiOH(aq) b. HC₂H₂O₂(aq) + Ca(OH)₂(aq) c. HCl (aq) + Ba(OH)₂(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Sure here are the completed and balanced a...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Procedures Experiment 1: Standardize the Iodine Solution Part 1: Prepare the Materials Take a 100 mL volumetric flask from the Containers shelf and place it on the workbench. Take ascorbic acid from...
-
Procedures Experiment 1: Standardize the Iodine Solution Part 1: Prepare the Materials Take a 100 mL volumetric flask from the Containers shelf and place it on the workbench. Take ascorbic acid from...
-
First, complete and balance each of the equations below. Then, choosing among ethanol, hexane, and liquid ammonia, state which (there may be more than one) might be suitable solvents for each of...
-
Unlike affirmative action, diversity _ _ _ _ _ . a . can exist even if organizations do not take purposeful steps to create it b . is required by law for private employers with 5 0 or more employees...
-
Why are accountants so concerned about their organization having an efficient and effective internal control system?
-
This makes no sense at all, said Bill Sharp, president of Essex Company. We sold the same number of units this year as we did last year, yet our profits have more than doubled. Who made the goofthe...
-
Why is an ethical culture considered necessary for fraud prevention and deterrence?
-
Gannett Glass Company manufactures three types of safety plate glass: large, medium, and small. All three products have high demand. Thus, Gannett Glass is able to sell all the safety glass that it...
-
Two large charged sheets are placed a distance D apart. The top sheet has uniform sur- face charge density + while the bottom sheet has uniform charge density -20. The sheets are large compared to...
-
Write balanced complete ionic and net ionic equations for each acidbase reaction. a. HBr(aq) + NaOH(aq) b. HF(aq) + NaOH(aq) C. HCHO(aq) + RbOH(aq)
-
Write balanced molecular and net ionic equations for the reaction between nitric acid and calcium hydroxide.
-
Why is obtaining a private college education more expensive on average than obtaining a public college education?
-
Consider a toothpaste manufacturer wanting to offer quantity discounts to maximize their profit. Should the manufacturer offer lot-sizebased or volume-based quantity discounts and, at the same time,...
-
Use basic managerial levers to improve supply chain profitability.
-
Consider a cosmetic companys inventory. What would be the impact on safety inventory levels if customers brand preferences suddenly change as a result of a campaign discouraging the use of...
-
A price discount where the pricing schedule offers discounts based on the quantity ordered in a single lot is customer based. lot size based. supplier based. volume based.
-
________ is the ability of a supply chain to delay product differentiation or customization until closer to the time the product is sold. Specialization Centralization Postponement Aggregation
-
At the beginning of 2010, Shanklin Company issued 10-year bonds with a face value of $1,000,000 due on December 31, 2019. The company wants to accumulate a fund to retire these bonds at maturity by...
-
What are some of the possible sources of information about a company that could be used for determining the companys competitive stance?
-
Calculate the velocity in m/s of a 12-kg object if it has a kinetic energy of 15 Nm. The formula for kinetic energy is KE = mv 2 , where m = mass and v = velocity.
-
Calculate the velocity in m/s of a 175-g body if it has a kinetic energy of 212 m Nm. The formula for kinetic energy is KE = mv 2 , where m = mass and v = velocity.
-
Calculate the kinetic energy in ft-lb of a 1-slug mass if it has a velocity of 4 ft/s. The formula for kinetic energy is KE = mv 2 , where m = mass and v = velocity.
-
Suppose we have n jobs where job J; requires time t and has priority pi. Our problem is to design a schedule of jobs given by i, 12, ..., in to minimize the weighted sum of the completion times, that...
-
5. Unemployment insurance and permanent job loss. Workers sometimes lose their jobs due to international trade. These jobs are permanently gone, and many displaced workers have difficulty finding a...
-
2. You are evaluating various investment opportunities currently available and you have the fol- lowing information about five different well-diversified portfolios of risky assets. Interest rate ry...

Study smarter with the SolutionInn App


