Complete each hydrogenation reaction. catalyst a. CH=CH-CH3 + H b. CH3-CH-CH=CH + H T CH3 c. CH3CHC=CH,
Question:
Complete each hydrogenation reaction.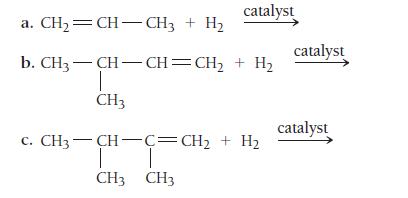
Transcribed Image Text:
catalyst a. CH₂=CH-CH3 + H₂ b. CH3-CH-CH=CH₂ + H₂ T CH3 c. CH3—CH—C=CH, + Hạ T T CH3 CH3 catalyst catalyst
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a CHCHCH3 HCH3CHCH3 ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Provide a figure showing the apparatus required to perform a hydrogenation reaction at 1atm. H2, without the need for specialist glassware/equipment. (The apparatus should be suitable for performing...
-
All chemists know that benzene is unusually stable, that is, it is aromatic. They are also well aware that many other similar molecules are stabilized by aromaticity to some extent and, more often...
-
Hydrogenation is an important chemical reaction of compounds that contain double bonds. Write a chemical equation for the hydrogenation of 1-hexene. Hydrogenation reactions are used extensively in...
-
Ava bought a house on 1 July 2000 for 62,000. She occupied the entire property as her PPR until 1 August 2007 when she began using one-quarter of the house for business purposes. This continued until...
-
The following information has been gathered for Leatroy Manufacturing: Net income ..................$5,000,000 Interest expense .................$400,000 Average total assets...
-
Beverly Mills has decided to lease a hybrid car to save on gasoline expenses and to do her part to help keep the environment clean. The car she selected is available from only one dealer in the local...
-
When conducting an incremental analysis, what step must always be taken immediately prior to beginning the pairwise comparisons? a. Order the alternatives from highest to lowest initial investment b....
-
Rodent Corporation produces two types of computer mice, wired and wireless. The wired mice are designed as low-cost, reliable input devices. The company only recently began producing the...
-
Let 1 f(z) = (2-1)(x-2) Find the Laurent expansion of f about z = 0 in the region {ze C: 1
-
Complete each hydrogenation reaction. a. CH3 CH,CH=CH, + H CH3 CH3 I catalyst b. CH3 -CH-C=C-CH3 + H c. CH3CH,C=CH, + H CH3 catalyst catalyst
-
What are the products of each alkene addition reaction? a. CH3 CH -CH=CH + Br 1 CH3 b.CH=CH-CH3 + Cl CH3 I C. CH3-C-CH=CH + HCI CH3 CH3 d. CH3 -CH-CH=C-CH3 + HBr T CH3-CH3
-
In Problems 4148, find an equation for the hyperbola described. Graph the equation. Vertices at (1, -3) and (1,1); asymptote the line y + 1 NIGE (x - 1)
-
1. One popular concept on stock market price is called "martingale pric- ing", which is relevant to "rational expectations" or "efficient market hypothesis" in economic theories. A rough statement on...
-
The workplace practice I chose to evaluate is, workplace culture described by employees as stressful, competitive, or alienating. For the reason why this ends up being the case I feel it starts with...
-
What will you do to make your objectives clear and candid during the conversation? How will you manage your emotion during the conversation while still maintaining empathy and trust with the...
-
If a rope loaded with a force of 100 lbf had a spring constant of 500 lbf/inch, how far would the rope stretch or elongate? 0.05" 3.14" Not Listed 0.005" 3.18" 5" 0.5"
-
The Amazon.ca logo has an arrow from the "a" to the "z," communicating that Amazon has everything from a to z. What is this an example of? Explain
-
Ritz-Carlton. The name alone evokes images of luxury and quality. As the first hotel company to win the Malcolm Baldrige National Quality Award, the Ritz treats quality as if it is the heartbeat of...
-
The cash records of Holly Company show the following four situations. 1. The June 30 bank reconciliation indicated that deposits in transit total $720. During July, the general ledger account Cash...
-
A series RLC circuit with R = 1.3 , L = 27 mH, and C = 0.33F is connected across a sine-wave generator. If the capacitors peak voltage rating is 600 V, whats the maximum safe value for the generators...
-
Differentiate Equation 28.9 to find the current in the LC circuit, and use q = CV to find the voltage. From these, obtain the electric energy in the capacitor and the magnetic energy in the inductor,...
-
Two capacitors are connected in parallel across a 24-V rms, 7.5-kHz sine-wave generator, and the generator supplies a total rms current of 56mA. With capacitors rewired in series, the rms current...
-
Your algorithm gets the following results in a classification experiment, where in the table, 'Id' is the index number, 'Prediction' is the predicted results, 'Target' is the ground truth that the...
-
Last month, 5 customers of Macland Automotive bought the extended service plan with their purchase of a new car. All of their credit scores are given below. Calculate the standard deviation of this...
-
5. (30pt) Write a 2-page report including a diagram of the resulting binomial tree in (a) and the capture and explanation of the results in (b). Maturity (year) 1 2 3 4 Yield (%) 9 9.5 10 10.5 Table...

Study smarter with the SolutionInn App


