Complete the table. a. 355 km/s b. 1228 g/L c. 554 mK/s d. 2.554 mg/mL _cm/s _g/mL
Question:
Complete the table.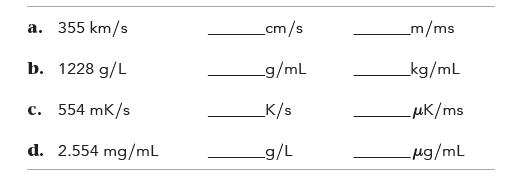
Transcribed Image Text:
a. 355 km/s b. 1228 g/L c. 554 mK/s d. 2.554 mg/mL _cm/s _g/mL _K/s _g/L _m/ms _kg/mL _μk/ms μg/mL
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The completed table is as follows Original unit Scientific notation Unit prefix ...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For an ideal gas, the decrease in internal energy of 1.4 kgm is -343KJ when the volume increases from 0.043 cu. m. to 0.13 from 0.07 bar to 0.02 bar; c, = 0.72 cu. m. and the pressure decreases kgm-K...
-
Complete the table below for contribution margin per unit, total contribution margin , and contribution margin ratio: A Number of units 1,720 units 14,920 units 4,620 units Sales price per unit $...
-
Problem 13-1A Calculation and analysis of trend percents LO A1, P1 Selected comparative financial statements of Haroun Company follow. HAROUN COMPANY Comparative Income Statements For Years Ended...
-
An electric field is E = 300 N/C i for x > 0 and E = 300 N/C i for x < 0. A cylinder of length 20 cm and radius 4 cm has its center at the origin and its axis along the x axis such that one end is at...
-
Andreasen Corporation manufactures thermostats for office buildings. The following is the cost of each unit: Materials . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
Partial comparative balance sheet and income statement information for Smith Company is as follows: In 2010, the year-end balances for Accounts Receivable and Inventory were $64,800 and $102,400,...
-
Journalize the following transactions of Cramer, Inc., which ends its accounting year on June 30: Apr 1 Jun 6 30 Loaned $20,000 cash to R. Simpson on a one-year, 8% note. Sold goods to Friday, Corp.,...
-
On January 1, 2017, Procise Corporation acquired 100 percent of the outstanding voting stock of GaugeRite Corporation for $1,980,000 cash. On the acquisition date, GaugeRite had the following balance...
-
By using database Trigger, how to answer this question? (given dreamhome tables) ii. Base on the Dreamhome database (refer appendix), implement the following data integrity rules: i. A property...
-
Write pseudocode for each example (a through e) in Exercise 2, making sure your pseudocode is structured and accomplishes the same tasks as the flowchart segment. Data From Exercise 2 a. b. D Yes NO...
-
Express the quantity 254,998 m in each unit. a. km b. Mm c. mm d. cm
-
Complete the table. a. 1245 kg b. 515 km C. 122.355 s d. 3.345 kJ 1.245 x 10 g _dm ms 1.245 x 10 mg _cm ks _mJ
-
To what extent are gains and losses on distributed assets recognized when C corporations, S corporations, or partnerships liquidate? Do the tax rules in this area always favor certain entities over...
-
Distinguish between the two main types of decisions, giving an example of each.
-
Describe the relationships depicted in an organizational chart.
-
Why is it important for a company to have a strong organizational culture?
-
Describe three different types of multidivisional structures. Do companies use only one of the types or can they be mixed?
-
What is the difference between departments and divisions? Can the terms be used interchangeably? Why or why not?
-
What are the objectives of the International Accounting Standards Committee Foundation, and does the FASB support those objectives?
-
How many years will it take a $700 balance to grow into $900 in an account earning 5%?
-
A compound with molecular formula C 5 H 11 N has no p bonds. Every carbon atom is connected to exactly two hydrogen atoms. Determine the structure of the compound.
-
For each of the following compounds, use the nitrogen rule to determine whether the molecular weight should be even or odd. Then calculate the expected m/z value for the molecular ion. a. b. c. d. O:...
-
Draw all constitutional isomers with molecular formula C 2 H 6 S, and rank them in terms of increasing acidity.
-
Discuss with suitable examples why activity-based costing (ABC) is better than the traditional costing system. Provide a suitable numerical example of ABC in the manufacturing sector and show all the...
-
Requirements: 1) Complete adjusted trial balance of worksheet 2) Use your completed worksheet to prepare (a) multi-step or single-step net income statement, (b) statement of owner's equity and (c) a...
-
$50,000 in sales for each of the months of April, May and June. You are to project an increase of 15% for each quarter of each liquor thereafter. Paul assures you that the current distillery...

Study smarter with the SolutionInn App


