For each of the following compounds, use the nitrogen rule to determine whether the molecular weight should
Question:
a.
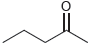
b.
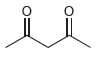
c.
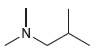
d.
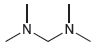
Transcribed Image Text:
O: O:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
a This compound does not have any nitrogen atoms Acco...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write the chemical formula for each of the following compounds, and indicate the oxidation state of nitrogen in each: (a) Sodium nitrite (b) Ammonia (c) Nitrous oxide (d) Sodium cyanide (e) Nitric...
-
For each of the following compounds, draw the important resonance forms. Indicate which structures are major and minor contributors or whether they have the same energy. (a) H2CNN (b) H2C=CH-NO2 (c)...
-
For each of the following compounds and ions, 1. Draw a Lewis structure. 2. Show the kinds of orbitals that overlap to form each bond. 3. Give approximate bond angles around each atom except...
-
Sedona Company set the following standard costs for one unit of its product for 2015. Direct material (20 Ibs. @ $2.50 per Ib.) . . . . . . . . . . . . . . . . . . . . . . . $ 50 Direct labor (10...
-
Implement the contains routine for MyLinkedList.
-
Most people in this country obtain health insurance through employer-sponsored plans. Although the historical background you just read explains how this system came about, it does not discuss whether...
-
The Signet Corporation management is transferring securities across their held-to maturity, trading, and available-for-sale portfolios. Within the industry, management has the reputation of being...
-
A partially completed worksheet for Home Auto Detailing Service, a firm that details cars and vans, follows on page 178. INSTRUCTIONS 1. Record balances as of December 31 in the ledger accounts. 2....
-
1.2. Complete the secondary allocation statement below (where applicable, round of figures to two decimal places). (6 marks) Production departments Service departments A B C X Y Total primary...
-
What was Lakesides gross profit for January? a. $7,500 b. $ 0 c. $1,900 d. $5,600
-
Economists call the reduction in consumer confidence that resulted from the September 11 th attacks an _____ shock which leads to the ________. a. Aggregate demand, aggregate demand curve shifting...
-
For developed economies sustained increases in aggregate supply, absent increases in aggregate demand, will result in a. Growth for a while, but ultimately it will result in only inflation. b....
-
Avery Grocery Store is the only grocery store in Rayville, a small town populated mostly by college students. Students have been complaining lately because Avery always runs out of macaroni and...
-
2. A person has completed a pre-license course of 63 classroom hours, 50 minutes each. At the end of the course she scored an 85% on the test and she has filed the appropriate fees and paperwork to...
-
The charter of a corporation provides for the issuance of 125,000 shares of common stock. Assume that 55,000 shares were originally issued and 13,400 were subsequently reacquired. What is the number...
-
Each of the following independent companies is missing numerical data. Required: Use your knowledge of the financial statement equations and their interrelationships to fill in the missing amounts....
-
1. How would you describe the case study organizations strategy in international perspective? 2. What are the organizations reasons for adopting such a strategy? 3. Do you think it is a feasible...
-
Alfred wants to describe the overall response to each of the 9 statements. Which statistic should he use? Alfred wants to compare the factor score distributions for the controllers and users...
-
Perform all indicated operations, and write each answer with positive integer exponents. p -1 - q -1 /)pq) -1
-
An Atomic Energy Commission nuclear facility was established in Hanford, Washington, in 1943. Over the years, a significant amount of strontium 90 and cesium 137 leaked into the Columbia River. In a...
-
What aromatic products would you obtain from the KMnO4 oxidation of the followingsubstances? (a) O2N. (b) C(CH3)3 CH(CH3)2 "
-
Refer to Table 5.3 for quantitative idea of the stability of a benzyl radical. How much more stable (in kJ/mol) is the benzyl radical than a primary alkyl radical? How does a benzyl radical compare...
-
Styrene, the simplest alkenylbenzene, is prepared commercially for use in plastics manufacture by catalytic dehydrogenation of ethylbenzene. How might you prepare styrene from benzene using reactions...
-
What might be challenges for public administrators in orchestrating ethical performances when working with individual ethics inside and outside the organization?
-
On January 1, 2023, Bertrand, Incorporated, paid $70,800 for a 40 percent interest in Chestnut Corporation's common stock. This Investee had assets with a book value of $235,000 and liabilities of...
-
In an airline's hiring process, all candidates were assessed on criteria such as "assertiveness," "teamwork" and "ability to have fun." Although these were age-neutral on their face, bias was...

Study smarter with the SolutionInn App


