Consider the molecular views of an Al strip and Cu 2+ solution. Draw a similar sketch showing
Question:
Consider the molecular views of an Al strip and Cu2+ solution.
Draw a similar sketch showing what happens to the atoms and ions after the Al strip is submerged in the solution for a few minutes.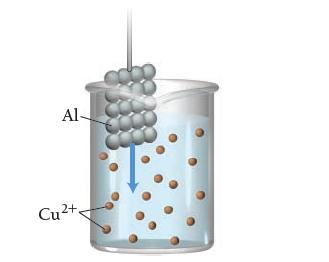
Transcribed Image Text:
Al Cu2+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The drawing should show that s...View the full answer

Answered By

SUMAN DINDA
I LIKE TO TEACH STUDENTS. SO, I START MYSELF AS A PRIVATE TUTOR. I TEACH STUDENTS OF DIFFERENT CLASSES. I HAVE ALSO DONE BACHELOR OF EDUCATION DEGREE(B.ED). DURING THIS COURSE I HAD TO TEACH IN A SCHOOL. SO I HAVE A GOOD EXPERIENCE IN TEACHING.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In Figure 1.19, arrows show the directions of the velocity and the acceleration of a ball moving in a circle. Make a similar sketch showing these directions for a car Figure 1.19 (a) speeding up from...
-
When a pure substance is placed in contact with water, there are three possible outcomes. The substance may do nothing that is, the substance does not dissolve and no visible change takes place. The...
-
One explanation for Chinas rapid economic growth during the past several decades is its expansion of policies that encourage technology transfer. By this, we mean policies such as opening up to...
-
Suppose you wanted to hasten the transition from a depletable fossil fuel to solar energy. Compare the effects of a per-unit tax on the depletable resource to an equivalent per-unit subsidy on solar...
-
Oaklawn National Bank has requested an analysis of checking account profitability by customer type. Customers are categorized according to the size of their account: low balances, medium balances,...
-
Imagine that Canada, the United States, and Mexico decide to adopt a fixed exchange rate system. What would be the likely consequences of such a system for (a) international businesses and (b) the...
-
Nationwide Insurance developed a program to recruit new insurance agents by offering planning, training, and start-up financing to build self-sustaining agencies. These new agents would be...
-
Bug-Off Exterminators provides pest control services and sells extermination products manufactured by other companies. The following six-column table contains the companys unadjusted trial balance as...
-
1. [10] Is Grtzsch graph M(C5) Hamiltonian? Is the complement of M(C5) Hamilto- nian? Justify your answer.
-
Consider the molecular view of an electrochemical cell involving the overall reaction: Draw a similar sketch of the cell after it has generated a substantial amount of electrical current. Zn(s) +...
-
Consider the unbalanced redox reaction: Balance the equation and determine the volume of a 0.850 M K 2 Cr 2 O 7 solution required to completely react with 5.25 g of Cu. 2+ CrO72 (aq) + Cu(s) Cr+(aq)...
-
How can the S&L crisis be blamed on the principal-agent problem?
-
Describe Walmart's international strategy leading up to its acquisition of Massmart in Africa. What did Walmart learn from its experiences in Germany, Japan, and South Korea? What grade would you...
-
How does a social development course provide insight into different aspects of communication, relationships, group dynamics, and human behavior?
-
Billy Bob has $1000. How much would he have after 8 years if he leaves it invested at 1.5% with annual compounding? (Answer in $s to the nearest cent, i.e. xxxx.xx with no $ sign needed.)
-
Describe thoroughly Herzberg's Two-Factor Model of Motivation (compare and contrast the two factors) and discuss at least three organizational features/concepts of both factors. Last, describe how...
-
Your investments increased in value by 15.6 percent last year but your purchasing power increased by only 11.3 percent. What was the approximate inflation rate? (Enter your answer as a percent...
-
Lake Charles Seafood makes 500 wooden packing boxes for fresh seafood per day, working in two 10-hour shifts. Due to increased demand, plant managers have decided to operate three 8-hour shifts...
-
Respond to the ethical judgments required based on the following scenarios. Scenario 1. Assume you have collected a sample using MUS and that you have evaluated that sample to calculate a total...
-
In the dangerous situation of prompt criticality in a fission reactor, the generation time drops to 100 s as prompt neutrons sustain the chain reaction. If a reactor goes prompt critical with k =...
-
How much heavy water (deuterium oxide, 2 H 2 O or D 2 O) would be needed to fuel a 1000-MW D-D fusion power plant for 1 year?
-
The protonproton cycle consumes four protons while producing 27 MeV of energy. (a) At what rate must the Sun consume protons to produce its power output of 4x10 26 W? (b) The present phase of the...
-
6 - 6 is The slope of the line y=5x- (Simplify your answer.)
-
You plan to invest $2,000 per year into a retirement account. If you earn a compound annual rate of return of 5%, how many years will it take you to reach a balance of $500,000? 50.14 47.22 51.99...
-
1. When applied to an equilibrium system, which of the following stresses would cause a change in the Ko value after the equilibrium has been re-established? a. Addition of catalyst b. Decrease in...

Study smarter with the SolutionInn App


