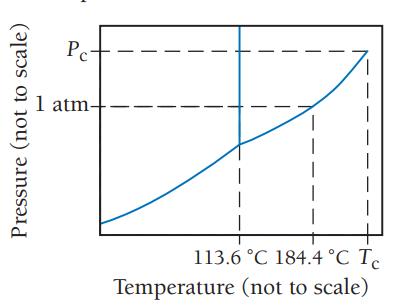
Consider the phase diagram for iodine shown here. a. What is the normal boiling point for iodine?
Question:
Consider the phase diagram for iodine shown here.
a. What is the normal boiling point for iodine?
b. What is the melting point for iodine at 1 atm?
c. What state is present at room temperature and normal atmospheric pressure?
d. What state is present at 186 °C and 1.0 atm?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
a Iodine normally boils at 1844C This is the temper...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In Problems 1956, solve each system of equations. If the system has no solution, state that it is inconsistent. For Problems 1930, graph the lines of the system. - - -2= = 1 -x + 2y - 3z = -4 3x-2y -...
-
Find the derivative of the function: f(x) = -4e-11x
-
Use powers of adjacency matrices to determine the number of paths of the specified length between the given vertices. Exercise 55, length 2, v 1 to v 3 Data From Exercise 55 0 1 0 0
-
A physical pendulum of mass m = 3 . 3 6 kg is comprised of an odd shape that has a centre - of - mass a distance of d = 0 . 5 5 5 m from the pivot point. The pendulum is displaced from equilibrium to...
-
On February 10, Mrs. Sunderhaus purchased a diamond ring from Perel & Lowenstein for $6,990. She was told by the companys salesperson that the ring was worth its purchase price, and she also received...
-
Barry Hannah, CPA, plans to use attributes estimation to help assess control risk for the Oxford Company's control procedures over credit sales transactions. Hannah has begun to outline main steps in...
-
A bar is \(4 \mathrm{~m}\) long and has a diameter of \(30 \mathrm{~mm}\). Determine the total amount of elastic energy that it can absorb from an impact loading if (a) it is made of steel for which...
-
Carlsville Company, which began operations in 2013, invests its idle cash in trading securities. The following transactions are from its short-term investments in its trading securities. 2013 Jan. 20...
-
Below are the financial statements for Patterson Suits for December 31, 2021, and some additional information. Based on the information provided, prepare the Statement of Cash Flows. (22 marks)...
-
Argon has a normal boiling point of 87.2 K and a melting point (at 1 atm) of 84.1 K. Its critical temperature is 150.8 K, and its critical pressure is 48.3 atm. It has a triple point at 83.7 K and...
-
Consider the phase diagram shown here. Identify the states present at points a through g. Pressure (not to scale) Pc- a. e 0Q g b G 14 Pl I Ic Temperature (not to scale)
-
In Problems, replace each radical with a fractional exponent. Do not simplify. 5 x 3
-
Geely Holding Group Acquires Volvo Cars: Chinese Private Enterprises Chase Their Dreams Overseas (text) Question 1. What opportunities and challenges did Geely Holding Group face when it acquired...
-
Sultan Company uses an activity-based costing system. At the beginning of the year, the company made the following estimates of cost and activity for its five activity cost pools: Activity Cost Pool...
-
How do chaperone proteins facilitate proper protein folding in the endoplasmic reticulum? What mechanisms exist to address misfolded proteins and prevent cellular stress?
-
What are the key checkpoints in the cell cycle, and what role do cyclins and cyclin-dependent kinases play in cell cycle progression and regulation ?
-
Camnet Industry is considering introducing a new line of higher end produces. Below are the company's forecast of financial data for the new products. Price per unit: $15 Quantity: ...
-
Overnight Laundry is considering the purchase of a new pressing machine that would cost $100,000 and would produce incremental cash flows of $25,000 annually for 6 years. The machine has a terminal...
-
An investor sells a European call on a share for $4. The stock price is $47 and the strike price is $50. Under what circumstances does the investor make a profit? Under what circumstances will the...
-
Calculate the rotational partition function for oxygen (B = 1.44 cm 1 ) at its boiling point, 90.2 K, using the high-temperature approximation and by discrete summation. Why should only odd values of...
-
In microwave spectroscopy a traditional unit for the rotational constant is the Mc or mega cycle equal to 10 6 s 1 . For 14 N 14 N 16 O the rotational constant is 12,561.66 Mc. a. Convert the above...
-
a. Calculate the percent population of the first 10 rotational energy levels for HBr (B = 8.46 cm 1 ) at 298 K. b. Repeat this calculation for HF assuming that the bond length of this molecule is...
-
IMPEL Co. 2022 and 2021 balance sheets showed the following items: December 31 2022 2021 Debits: Cash $ 27,000 $ 21,000 Accounts receivable 43,000 49,000 Merchandise 113,000 97,000 inventory...
-
Factor out the indicated common factor. -6y+10y+8 Part 1 of 2 Factor out 2. 2 -6y + 10y+ 8 =
-
Find standard deviation here. Episodes of Illness Number of Episodes of Illness Number of Patients 0 1 54754 2 2 1 234567 Calculate n, (X), and X. n = 8 () = 30 X = n = ?

Study smarter with the SolutionInn App


