Consider the phase diagram shown here. Identify the states present at points a through g. Pressure (not
Question:
Consider the phase diagram shown here. Identify the states present at points a through g.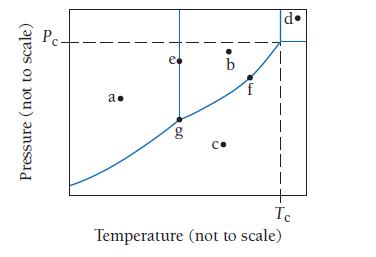
Transcribed Image Text:
Pressure (not to scale) Pc- a. e 0Q g b G 14 •Pl I Ic Temperature (not to scale)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a Solid b Liquid ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A particular liquid crystalline substance has the phase diagram shown in the figure. By analogy with the phase diagram for a nonliquid crystalline substance, identify the phase present in each area. T
-
5. Consider the phase diagram for iodine and answer each question. a. What is the normal boiling point for iodine? b. What is the melting point for iodine at 1 atm? c. What state is present at room...
-
Given the definition of the radian angle: S l Where [s] = m, [4] = m What are the dimensions of angles? A. No way to know B. meters, m C. 1 (or no units/dimensions) D. seconds, S E. kilograms, kg
-
Iverson owned Iverson Motor Company, an enterprise engaged in the repair as well as the sale of Oldsmobile, Rambler, and International Harvester Scout automobiles. Forty percent of the businesss...
-
Which of the following amino acids are more likely to be found on the outside of a globular protein, and which on the inside? Explain. (a) Valine (b) Aspartic acid (c) Phenylalanine (d) Lysine
-
After returning from a skiing vacation in Vermont, Leslie Adel came down with Legionnaires Disease. He claimed it was from the water drunk at the ski resort and provided by Greensprings of Vermont...
-
Sharp Company manufactures a product for which the following standards have been set: During March, the company purchased direct materials at a cost of $55,650, all of which were used in the...
-
4. Consider the tables of values for the two functions shown. x y = f(x) -2 5 y = g(x) -2 -1 6 -1 0 8 1 1 7 0 2 9 2 Complete the table of values for the composite function y = f(g(x)). - x y =...
-
Consider the phase diagram for iodine shown here. a. What is the normal boiling point for iodine? b. What is the melting point for iodine at 1 atm? c. What state is present at room temperature and...
-
How much heat (in kJ) is evolved in converting 1.00 mol of steam at 145 C to ice at -50 C? The heat capacity of steam is 2.01 J/g C, and that of ice is 2.09 J/g C.
-
Construct phrase-structure grammars to generate each of these sets. a) {0n | n 0} b) {1n0 | n 0} c) {(000)n | n 0}
-
You are the HR Director of AAA insurance company and until now you have taken a traditional approach to recruitment and selection. However, you have been noticing modern trends around the use of...
-
Assume you established a new pastry shop. 1- How would you segment your customers (What may be the effective segmentation criteria?) 2- What segment would you target? Why? 3- How would you position...
-
LaToya Limmons is the manager of the Human Resources (HR) department and Javier Jartin is the manager of the sales department for a production company headquartered in the United States but also has...
-
Task details: You are to write a Reflective Journal on: 1. how the readings, class activities, discussions observations and lectures have changed how you communicate at work or life 2. the things...
-
You are preparing to leave home for several days. You cannot bring along your pets and your houseplants, and they will both need special care because they are alive. How is a living organism...
-
[This is a variation of Exercise 714 modified to focus on a noninterest-bearing note.] On June 30, 2013, the Esquire Company sold some merchandise to a customer for $30,000 and agreed to accept as...
-
A bar of a steel alloy that exhibits the stress-strain behavior shown in Figure 6.22 is subjected to a tensile load; the specimen is 375 mm (14.8 in.) long and has a square cross section 5.5 mm (0.22...
-
In general, the high-temperature limit for the rotational partition function is appropriate for almost all molecules at temperatures above their boiling point. Hydrogen is an exception to this...
-
When 4 He is cooled below 2.17 K it becomes a superfluid with unique properties such as a viscosity approaching zero. One way to learn about the superfluid environment is to measure the...
-
Calculate the vibrational partition function for H 35 Cl ( = 2990 cm 1 ) at 300 and 3000. K. What fraction of molecules will be in the ground vibrational state at these temperatures?
-
Use source code: Header file: Main file: In the main program, add lines of code to perform the following on a dynamically allocated single instance of the class: a) Use new to create a single...
-
1. WHICH IS THE LATEST VERSION OF MICROSOFT EXCEL AVAILABLE FOR WINDOWS COMPUTERS. a) 2013 b) 2022 c) 2023 d) 2024 2. WHAT IS THE EQUIVALENT OF A PAPER NOTEBOOK IN EXCEL. a) WORKBOOK b) WORKSHEET c)...
-
All instructions/questions are referring to one code, please anwser them all Use this source code as a guide: Code the following: a) Allocate the array in main at the location indicated by the...

Study smarter with the SolutionInn App


