Consider the structure of the amino acid alanine. Indicate the hybridization about each interior atom. H
Question:
Consider the structure of the amino acid alanine. Indicate the hybridization about each interior atom.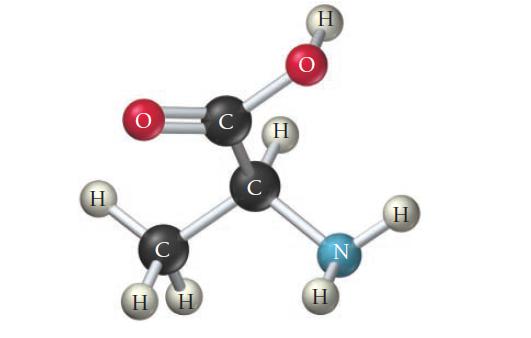
Transcribed Image Text:
H Η Η Η H H H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
H H H ...View the full answer

Answered By

Mary Boke
As an online tutor with over seven years of experience and a PhD in Education, I have had the opportunity to work with a wide range of students from diverse backgrounds. My experience in education has allowed me to develop a deep understanding of how students learn and the various approaches that can be used to facilitate their learning. I believe in creating a positive and inclusive learning environment that encourages students to ask questions and engage with the material. I work closely with my students to understand their individual learning styles, strengths, and challenges to tailor my approach accordingly. I also place a strong emphasis on building strong relationships with my students, which fosters trust and creates a supportive learning environment. Overall, my goal as an online tutor is to help students achieve their academic goals and develop a lifelong love of learning. I believe that education is a transformative experience that has the power to change lives, and I am committed to helping my students realize their full potential.
5.00+
4+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Suppose you are the head of the marketing department for an on-demand video streaming service. A registered user pays a monthly subscription fee to access the platforms entire library. Your research...
-
Consider the structure of the amino acid aspartic acid. Indicate the hybridization about each interior atom. H 0 0 Z H H H H
-
Write a hybridization and bonding scheme for each molecule that contains more than one interior atom. Indicate the hybridization about each interior atom. Sketch the structure, including overlapping...
-
. 4. Bank overdrafts repayable on * 1 point demand may be included in the cash and cash equivalent balance. True O False
-
Dilts Corporation sells three different models of mosquito zapper. Model A12 sells for $50 and has variable costs of $40. Model B22 sells for $100 and has variable costs of $70. Model C124 sells for...
-
Amy Van Dyken Construction Company uses the percentage-of-completion method of accounting. In 2008, Van Dyken began work under contract #E2-D2, which provided for a contract price of $2,200,000....
-
Gold-on-gold nuclear collisions at the Relativistic Heavy Ion Collider (RHIC) at the Brookhaven National Laboratory create a quark-gluon plasma with an energy density of about \(4 \mathrm{GeV} /...
-
In the early 1980s, New England Electric System (NEES) was deciding how much to bid for the salvage rights to a grounded ship, the SS Kuniang. If the bid were successful, the ship could be repaired...
-
Karen wishes to have $19,526 cash for a new car 5 years from now. How much should be placed in an account now, if the account pays 5.5% annual interest rate, compounded weekly?
-
Sketch the bonding molecular orbital that results from the linear combination of two 1s orbitals. Indicate the region where interference occurs and state the kind of interference (constructive or...
-
Write a hybridization and bonding scheme for each molecule or ion. Sketch the structure, including overlapping orbitals, and label all bonds using the notation shown in Examples 11.6 and 11.7. a. SO...
-
Iris sighed as she completed her initial review of her staffs checklist results. She pulled out a notepad and began outlining the projects she foresaw, based on the shortcomings identified via the...
-
Paul, a software designer, has decided to submit a proposal with a local school. In his proposal's introduction, he states that there's new software available to maintain and track a student's...
-
Explain essential terms and material terms terms in the real estate environment.
-
For the bolted connection shown in Figure, Compute the design strength ftPn, neglecting block shear. The angle is made of A36 steel (fy-36 ksi, fu-58 ksi) The bolts are 1 in diameter. Assume U=0.9 +...
-
Sound waves create a physiological response in which the brain interprets the sound waves and correlates those with a meaning. What is this passive process known as?
-
What is the thesis of the essay Free Minds and Hearts at work? What support to the thesis? And why agree or disagree? Explain
-
You have worked as a real estate agent for 10 years and are earning about $100,000 per year with your current agency. You prepared the following information to use in evaluating the financial...
-
The electric field due to a line charge is given by where l is a constant. Show that E is solenoidal. Show that it is also conservative. E =
-
Referring back to the multiple antenna system on p. 456, compute the angular separation between successive lobes or principal maxima and the width of the central maximum.
-
Examine the setup of Fig. 10.3 in order to determine what is happening in the image space of the lenses; in other words, locate the exit pupil and relate it to the diffraction process. Show that the...
-
Consider the case of single-slit Fraunhofer diffraction. Calculate the ratio of the irradiance of the central maximum to the irradiance of the first secondary maximum on either side of it. Check your...
-
The financial and economic decisions hospitals face in our current healthcare system.Explain how cost-shifting impacts the flow of hospital monies. Describe "cream skimming" and determine if it is an...
-
op oil exporter Saudi Arabia is expected to hike its prices for all crude grades it sells to Asia-Pacific in November/December 2019 after an attack on its oil facilities led to a spike in Middle East...
-
A country is facing a recesionnary gap Graph the scenario Suggest a monetary policy and explain all the steps ( bonds market, exchange rate, interest rate) Show the change on the graph of Q1 What is...

Study smarter with the SolutionInn App


