Consider the structure of the amino acid aspartic acid. Indicate the hybridization about each interior atom. H
Question:
Consider the structure of the amino acid aspartic acid. Indicate the hybridization about each interior atom.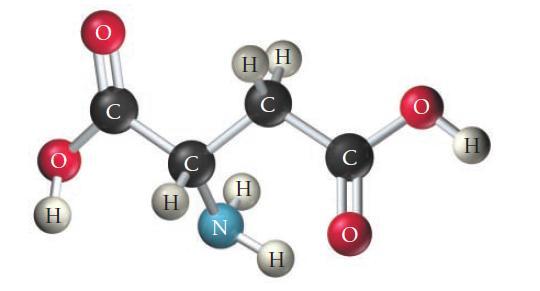
Transcribed Image Text:
H 0 0 Z H H H H О
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
OHCOCH2CNH2COOH this is the structure of aspartic acid The hybridization of c...View the full answer

Answered By

Jeetu sahu
I completed my B.Tech in Textile Engineering from NIT JALANDHAR,INDIA. I can help students to solve assignments and exams of physics ,mathematics, statistics, science and engineering. I am doing it since three years. I can provide tuition for Physics and Mathematics .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Suppose you are the head of the marketing department for an on-demand video streaming service. A registered user pays a monthly subscription fee to access the platforms entire library. Your research...
-
Consider the structure of the amino acid alanine. Indicate the hybridization about each interior atom. H H H H
-
Write a hybridization and bonding scheme for each molecule that contains more than one interior atom. Indicate the hybridization about each interior atom. Sketch the structure, including overlapping...
-
Hi,Please answer no.5 and 6 only. This is my 3rd time to uploadthis question. Thank you so much! Bay Lake Mining Ltd. purchases earth-moving equipment on 1 August \( 20 \times 6 \) and signs a...
-
Information for Dilts Corporation is given in BE19-7. If the company has fixed costs of $199,500, how many units of each model must the company sell in order to break even?
-
Yogi Bear Corporation sold equipment to Magilla Company for $20,000. The equipment is on Yogis books at a net amount of $14,000. Yogi collected $10,000 in 2007, $5,000 in 2008, and $5,000 in 2009. If...
-
Calculate the energy density versus temperature very early in the universe when the temperatures were above \(k T=300 \mathrm{MeV}\). At those temperatures, quarks and gluons were released from...
-
Congratulations! Youve won a state lotto! The state lottery offers you the following (after- tax) payout options: Option # 1: $ 15,000,000 four years from now Option # 2: $ 2,200,000 at the end of...
-
1. Using Fig 2.16 calculate S max of solar constant (irradiance) corresponding to the maximum irradiance over the 25 years presented on the graph (round your answer to 4 digits). 2. Calculate RF max...
-
Sketch the bonding molecular orbital that results from the linear combination of two 1s orbitals. Indicate the region where interference occurs and state the kind of interference (constructive or...
-
Write a hybridization and bonding scheme for each molecule or ion. Sketch the structure, including overlapping orbitals, and label all bonds using the notation shown in Examples 11.6 and 11.7. a. SO...
-
On Earth where a = -9.8 m/s2 Suppose an object is thrown from a height of h = 100 m with velocity v = 5.0 m/s (upward) to find its trajectory in a local gravitational field of strength a. For the...
-
Human Communication Theory essay question: Describe cultivation theory and the important aspects of it. Give an example of it as well. Also, describe Cultivation differential.
-
Use the life details below that are appropriate for last name starting initial to answer the below questions. (QS 1) Estimate your expected weekly budget that you would need for food and...
-
The Colonial Pipeline is one of the most important gasoline pipelines in the United States; it provides over half of the petroleum that is used on the East Coast. The pipeline was knocked down in May...
-
The eleven person team at Albatross Gaming, LLC was faced with a confluence of several pleasant, yet complex, and some mutually exclusive, decisions. After seven years of the entire team subsisting...
-
Why is understanding the difference between rate of talk and rate at which brain processes language important as a speaker? 1: What does listening have to do with being an effective presenter?
-
The Plymouth Software Company has the following demand curve with MC = $10 and P = 100 Q with MR = 100 2Q. The company has option of charging monopolist price or perfect competitor price. Here it...
-
Show that every group G with identity e and such that x * x = e for all x G is abelian.
-
What does the term ISA mean? Do the Pentium 4 -> IBM POWER7 and Pentium 3 -> IBM POWER8 have the same ISA?
-
The diameter of an atom is roughly 104 times the diameter of its nucleus. If the nucleus of an atom were 1 mm across, how many feet across would the atom be?
-
In Section 10.1.3 we talked about introducing an intrinsic phase shift ε between oscillators in a linear array. With this in mind, show that Eq. (10.18) becomes when the incident plane...
-
13. Consider the large open economy model. Due to economic uncertainty, American investors decide to invest more in Canada. What happens to the real interest rate, the exchange rate, net capital...
-
A new heat exchanger may be purchased for 28,500$. At the end of 10 years it will be worth 33,000$ given i=12% what uniform annual benefit (EUAB) IS REQUIRED TO JUSTIFY THIS PURCHASE? PROVIDE cash...
-
A firm has demand equation Q = 30 - 5P . The firm must sell an integer quantity of product and charge the same price per unit of product for all units sold. It costs the firm $4.50 to produce and...

Study smarter with the SolutionInn App


