Consider the three generic weak acids HA, HB, and HC. The images shown here represent the ionization
Question:
Consider the three generic weak acids HA, HB, and HC. The images shown here represent the ionization of each acid at room temperature. Which acid has the largest Ka?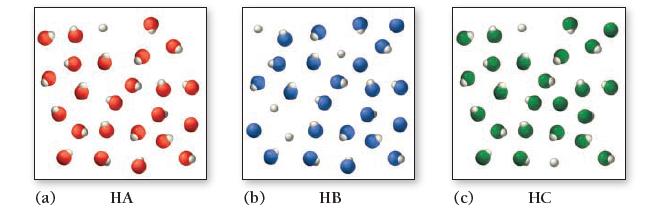
Transcribed Image Text:
(a) HA (b) HB (c) HC
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
b HB...View the full answer

Answered By

Douglas Makokha
Unlock Academic Success with Dedicated Tutoring and Expert Writing Support!
Are you ready to excel in your academics? Look no further! As a passionate tutor, I believe that dedication and hard work are the keys to achieving outstanding results. When it comes to academics, I strive to provide nothing but the best for every student I encounter.
With a relentless thirst for knowledge, I have extensively researched numerous subjects and topics, equipping myself with a treasure trove of answers to tackle any question that comes my way. With four years of invaluable experience, I have mastered the art of unraveling even the most intricate problems. Collaborating with esteemed writers has granted me exclusive access to the trade secrets utilized by the industry's top professionals.
Allow me the pleasure of assisting you with your writing assignments. I thrive on challenges and will guide you through any obstacles you may face. Together, we will unlock your academic potential and pave the way for your success.
4.90+
60+ Reviews
338+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
The diagrams here show three weak acids HA (A = X, Y, or Z) in solution. (a) Arrange the acids in order of increasing Ka. (b) Arrange the conjugate bases in increasing order of Kb. (c) Calculate the...
-
Consider the acids in Table. Which acid would be the best choice for preparing a pH = 7.00 buffer? Explain how to make 1.0 L of this buffer. Table Values of Ka for Some Common Monoprotio Acids...
-
Photons of wavelength 1.68 cm excite transitions from the rotational ground state to the first rotational excited state in a gas. Whats the rotational inertia of the gas molecules?
-
The theoretical cycle time for a product is 48 minutes per unit. The budgeted conversion costs for the manufacturing cell dedicated to the product are $4,320,000 per year. The total labor minutes...
-
Analog proportional-derivative controllers sometimes are formulated with a transfer function of the form: where ? = 0.05 to 02. The ideal PD transfer function is obtained when ? = 0. G1(s) = Kc(?DS +...
-
In 2001, the City of New York and the Dormitory Authority of the State of New York (DASNY) entered into an agreement to build a forensic biology laboratory in Manhattan. Per the agreement, DASNY...
-
Bunker Hill Mining Company has two competing proposals: a processing mill and an electric shovel. Both pieces of equipment have an initial investment of $ 750,000. The net cash flows estimated for...
-
In June 2022, the Federal Reserved imposed its first 0.75% interest rate hike -- the largest increase since 1994. Starting in January 2023, they dropped back to lower rate increases, but the rate...
-
Consider the given acid ionization constants. Identify the strongest conjugate base. Acid HNO(aq) HCHO(aq) HCIO(aq) HCN(aq) a) NO (aq) c) CIO (aq) Ka 4.6 x 10-4 1.8 x 10-4 2.9 X 10 8 4.9 10-10 b)...
-
Calculate the pH of each solution at 25 C and indicate whether the solution is acidic or basic. (a) [H3O+] 1.8 x 10-4 M = (b) [OH-] = 1.3 x 10-2 M
-
The following output (from \(\mathrm{R}\) ) presents the results of a hypothesis test for a population mean \(\mu\). a. Is this a one-tailed or a two-tailed test? b. What is the null hypothesis? c....
-
Which of the following are signs of inflation? a. The price of a house in a high-demand market increased by 6% last year. b. CPI in the European Union was 106 in 2020 and 109 in 2021. c. The price of...
-
An economic downturn throws millions of people out of work. In some industries, workers who remain employed, or insiders, continue to develop their skills, enabling them to push wages above the level...
-
Javier is a department manager at a big box store. Over the last month sales have slumped and he has lots of inventory going unsold. Now its time to put in his orders to restock for next month. How...
-
You take out student loans to help pay for your degree at a 5% annual interest rate. Assume the bank expected inflation to average 3% per year. What real interest rate did they expect to earn from...
-
Both CPI and the GDP deflator measure the change in the price of goods and services and tend to change in similar ways over time. In 2021, CPI rose by 4.7%, while the GDP deflator rose by 4.1%....
-
Journ Co. purchased short- term investments in available- for- sale securities at a cost of $ 50,000 on November 25, 2013. At December 31, 2013, these securities had a fair value of $ 47,000. This is...
-
It is possible to investigate the thermo chemical properties of hydrocarbons with molecular modeling methods. (a) Use electronic structure software to predict cHo values for the alkanes methane...
-
The vision of a hyperope is corrected with a +9D spectacle lens worn 12 mm from the cornea. Determine the appropriate power of a replacement contact lens.
-
We wish to correct the vision of a 7D my ope, whose both eyes are the same, with spectacles worn 15 mm from the eye. Determine the appropriate power.
-
An optometrist finds that a farsighted person has a near point at 125 cm. What power will be required for contact lenses if they are effectively to move that point inward to a more workable distance...
-
Stream Company plans to own and operate a storage rental facility. For the first month of operations, the company has the following transactions. 1. January 1 Issue 10,000 shares of common stock in...
-
On January 1, 2021, Tennessee Harvester Corporation issued debenture bonds that pay interest semiannually on June 30 and December 31. Portions of the bond amortization schedule appear below: Payment...
-
Edison Leasing leased high-tech electronic equipment to Manufacturers Southern on January 1, 2024. Edison purchased the equipment from International Machines at a cost of $123,288. Note: Use tables,...

Study smarter with the SolutionInn App


