Determine the formal charge of nitrogen in the structure shown here: a) +1 H: H-C-N-8: b) +2
Question:
Determine the formal charge of nitrogen in the structure shown here: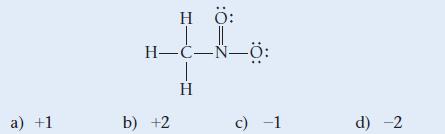
Transcribed Image Text:
a) +1 HÖ: H-C-N-8: b) +2 H c) -1 d) -2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

Murshidha parveen
I have a degree in Business Administration from the National University of Córdoba. I've been working as an online tutor for the past four years. I specialize in history and business articles. Every day I help students to do their homework, complete projects, dispel doubts, and much more. I hope to be a force in helping you get the knowledge you need.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In N 2 O, nitrogen is the central atom, and the oxygen atom is terminal. In OF 2 , however, oxygen is the central atom. Use formal charges to explain why. Formal charge = number of valence electrons...
-
Consider the Lewis structure for the polyatomic oxyanion shown here, where X is an element from the third period (Na --- Ar). By changing the overall charge, n, from 1 -- to 2 --- to 3 --- we get...
-
Identify the tort or possible tort claim. Defamation Libel Describe the essential elements of the tort or possible tort claim. These elements are provided by law. It is not necessary in response to...
-
In its first year of operation, Oriole Printing Shop estimated manufacturing overhead costs and activity in order to determine a predetermined overhead rate. At year end, March 31, overhead was...
-
Sonne Company produces a perfume called Whim. The direct materials and direct labor standards for one bottle of Whim are given below: During the most recent month, the following activity was...
-
Derive an integral expression for H(z) inside the unit circle in terms of Re{H(e j )} when h[n] is a real, stable sequence such that h[n] = 0 for n > 0.
-
On February 20, 2009, Cedar Valley Aviation, a wholly owned subsidiary of Aerial Services, Inc. (ASI), brought a Piper 522AS (Cheyenne II) in for maintenance to Des Moines Flying Service, Inc....
-
Jasmine Park encountered her boss, Rick Gompers, at the pop machine in the lobby. Rick is the vice president of marketing at Down South Lures Corporation. Jasmine was puzzled by some calculations she...
-
Consider the closed system with mass-action rate constants as shown. Suppose the rate constants are (in min-1) k = 0.05, kg = 0.7, k2= 0.005, and k4 = 0.4. ki k3 A B C k2 ka (a) Construct a...
-
Write the Lewis structure for XeF 2 .
-
Why is the formation of solid sodium chloride from solid sodium and gaseous chlorine exothermic, even though it takes more energy to form the Na + ion than the amount of energy released upon...
-
Nine-year-old Talia is afraid to hug and kiss her grandmother, who has cancer. What explains Talia's mistaken belief that the same behaviors that cause colds to spread might lead her to catch cancer?...
-
Suppose that all wages and prices in an economy are indexed to inflation that is, wages and prices are automatically adjusted to incorporate the latest inflation figures. Can there still be an...
-
Discuss how the availability of data creates interesting potential for research investigations.
-
In January 2020, the Boeing 777X, an update to the widely popular 777, took its maiden flight. The 777X was the product of what Boeing calls its advanced manufacturing process. With it, Boeing...
-
Discuss the respective roles of the SEC and the FASB in the development of accounting regulation and in determining how a company reports annually.
-
Sir Richard Branson, the billionaire founder of Virgin Group, has told 170 staff working for its head office that they can take as much annual leave as they like, providing it will not damage the...
-
On January 1, 2010, the Stimpson Company sells land to Barker Company for $2.5 million, then immediately leases it back. The relevant information is as follows: 1. The land was carried on Stimpsons...
-
Based on the scenario described below, generate all possible association rules with values for confidence, support (for dependent), and lift. Submit your solutions in a Word document (name it...
-
An A-36-steel hoop has an inner diameter of 23.99 in., thickness of 0.25 in., and width of 1 in. If it and the 24-in.-diameter rigid cylinder have a temperature of 65° F, determine the...
-
A pressure-vessel head is fabricated by welding the circular plate to the end of the vessel as shown. If the vessel sustains an internal pressure of 450 kPa, determine the average shear stress in the...
-
The gas pipe line is supported every 20 ft by concrete piers and also lays on the ground. If there are rigid retainers at the piers that hold the pipe fixed, determine the longitudinal and hoop...
-
Given the following transition matrix, calculate the 2 year cumulative default probabilities of B rated bond and a C rated bond respectively. (10 marks) Starting Ending state Total State probability...
-
Laker Company reported the following January purchases and sales data for its only product. For specific identification, ending inventory consists of 275 units from the January 30 purchase, 5 units...
-
(CPA) Purvis Company manufactures a product that has a variable cost of $50 per unit. Fixed costs total $1,000,000 and are allocated on the basis of the number of units produced. Selling price is...
Real World Algebra Understanding The Power Of Mathematics 1st Edition - ISBN: 0967991528 - Free Book

Study smarter with the SolutionInn App


