In N 2 O, nitrogen is the central atom, and the oxygen atom is terminal. In OF
Question:
In N2O, nitrogen is the central atom, and the oxygen atom is terminal. In OF2, however, oxygen is the central atom. Use formal charges to explain why.
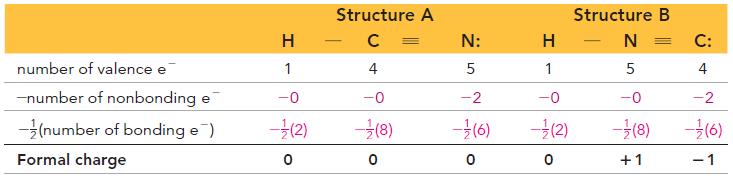
Transcribed Image Text:
Formal charge = number of valence electrons - (number of nonbonding electrons + number of bonding electrons) So the formal charge of hydrogen in HF is 0: Formal charge = 1 - [0+ (2)]=0 Number of valence electrons for H Number of electrons that H "owns" in the Lewis structure Similarly, we calculate the formal charge of fluorine in HF as 0: Formal charge = 7 - [6+ (2)]=0 Number of valence electrons for F Number of electrons that F "owns" in the Lewis structure
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
The question is asking us to use formal charges to explain why nitrogen is the central atom in N 2 O while oxygen is the central atom in OF 2 Formal c...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write a GUI application that records items of stock for a business. A barcode, description, stock level and price are recorded for each item. New items must be able to be added, and existing items...
-
Oxidation of the cyanide ion produces the stable cyanate ion (OCN2). The fulminate ion (CNO2), on the other hand, is very unstable. Fulminate salts explode when struck; Hg(CNO)2 is used in blasting...
-
Use the LE model to describe the bonding in H2CO and C2H2. Carbon is the central atom in H2CO, and C2H2 exists as HCCH.
-
L-keys, such as the one shown in the figure below, are commonly used to insert screws and bolts in medical implants. This insertion process can be simplified into the loading scenario shown in the...
-
What is the difference between sorting records and indexing records in a database?
-
Describe and discuss the saving-investment cycle.
-
Which interest rate on a bond determines the amount of the semiannual interest payment? a. Market rate b. Effective rate c. Stated rate d. Semiannual rate
-
Health experts recommend that runners drink 4 ounces of water every 15 minutes they run. Although handheld bottles work well for many types of runs, all-day cross-country runs require hip-mounted or...
-
1 F G K M R Question 1: On January 1st, 2020 Mandrake Company grants 5,000,000 shares of its $2 par common stock as restricted stock units for their employees. The market value on the grant date is...
-
How important is the resonance structure shown here to the overall structure of carbon dioxide? Explain. :0=C:
-
Use formal charge to identify the better Lewis structure. H H-C= H H=C
-
Saxton Corporation purchased 25 percent of Taylor Company's voting stock on January 1,2010, for $3 million in cash. At the date of acquisition, Taylor reported its total assets at $60 million and its...
-
Under the no arbitrage condition, when T increases, does the discount factor Z(0,T) necessarily increase or decrease? Why? When T increases, does the yield r(0,T) necessarily increase or decrease?...
-
Answer the questions based on the partially completed network diagram of a project below where the five values in each square grid represent the task time (center), ES (bottom left), EF (bottom...
-
Some fusion reactors use an induction process in which thermal neutrons collide with deuterium particles that are essentially twice as massive ( consisting of one proton and one neutron ) . Consider...
-
Q5. You are saving for junior's college is 18 years. You are saving $350 a month a you have secured an annual rate of 6%. How much will Junior have for college tuition? (Hint: Use PMT formula and...
-
Many investors believe the slope of yield curve is a leading economic indicator. Do you agree? Why? https://home.treasury.gov/policy-issues/financing-the-government/interest-rate-statistics?
-
Standard and actual material information is given in the following table for four companies. Required: For each company, calculate the missing ï¬gures. Assume that the quantity of material...
-
Suppose you need to answer any four of seven essay questions on a history test and you can answer them in any order. a. How many different question combinations are possible? b. What is the...
-
Why is mercury a convenient fluid to use in a barometer?
-
If water were to be used instead of mercury in a barometer, how high would the water column be?
-
What is the barometric pressure reading in inches of mercury corresponding to 14.696 psia?
-
2. For the last five years Joe has made deposits of $400 at the end of every six months earning interest at 3% compounded semi-annually. If he leaves the accumulated balance in an account earning 6%...
-
Calculate the present value of an annuity of $3,900 for four years, assuming an opportunity cost of 10%. To buy his favorite car, Larry is planning to accumulate money by investing his Christmas...
-
+ 3. Compute the PVBP, McCauley's Duration and Modified Duration for the following bonds. Settlement date is September 29, 2017. Explain why the duration differs (if any) for bonds with the same...

Study smarter with the SolutionInn App


