Determine the product of the reaction: CH3-CH-CH-OH CH3 a) CH3-CH - CH3 Cl b) CH3-CH - CH3
Question:
Determine the product of the reaction: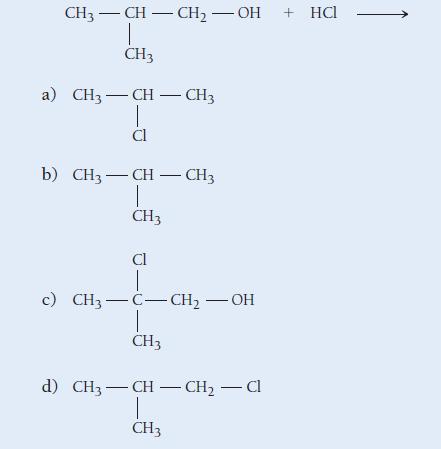
Transcribed Image Text:
CH3-CH-CH₂-OH CH3 a) CH3-CH - CH3 Cl b) CH3-CH - CH3 I CH3 CI c) CH3–C–CH, - OH CH3 d) CH3-CH-CH₂ - Cl | CH3 + HCl
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
d C...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Demonstrate your knowledge of palladium cross-coupling reactions by suggesting structures for compounds (14) and (15). Suggest reagent(s) that would facilitate the cross-coupling reaction between...
-
When (S)-1-bromo-1-fluoroethane reacts with sodium methoxide, an S N 2 reaction takes place in which the bromine atom is replaced by a methoxy group (OMe). The product of this reaction is...
-
Sodium-23 (in a sample of NaCl) is subjected to neutron bombardment in a nuclear reactor to produce 24 Na. When removed from the reactor, the sample is radioactive, with activity of 2.54 10 4 dpm....
-
Do you think firms rely too much on internal funds? Would it be better if they had to go back to shareholders when they needed cash?
-
Indicate whether each of the following actions is related to (a) Managing cash needs, (b) Setting credit policies, (c) Financing receivables, or (d) Ethically reporting receivables: 1. Selling...
-
The following expenditures were incurred during the year: 1. Paid $4,000 for an overhaul of an automobile engine. 2. Paid $20,000 to add capacity to a cellular phone companys wireless network. 3....
-
Which of the following is an activity not usually associated with forensic accounting and fraud examination consulting and litigation support? 1. A. Assessing fraud risk associated with internal...
-
Notes and Interest Glencoe Inc. operates with a June 30 year-end. During 2010, the following transactions occurred: a. January 1: Signed a one-year, 10% loan for $25,000. Interest and principal are...
-
Implement the Boolean function F(A.B.C.D) - (0,1,3,4,8,9,15) with a. a multiplexer b. a decoder Question 2 Draw the NAND gate representation of G A+ B'C' + DC. [10 marks] [5 marks] [5 marks]
-
Which compound is an ester? a) CH3 - CH -0 - CH3 O b) CH3-CH-C-OH c) CH3-C- CH3 O= d) CH3-C-0-CHCH3
-
Determine the product of the reaction: CH3 I CH3 -CH=C-CH3 + HBr CH3 T a) CHBr -CH=C-CH3 CH3 b) CH3 - CH -CH-CH3 CH3 c) CH3 -CH=CH-CH3 Br CH3 d) CH3 - CH -C-CH3 Br
-
In Example 9.13, the following finite distributed lag model was estimated for Okun's Law using the data file okun5_aus. a. Find the correlogram of the least squares residuals for this model. Is there...
-
Briefly describe the links between the Facility manager, Director of patient care, compliance officer, risk management, hospital staff, patients, the community, the CMS, and the joint commission in...
-
You are required to update the cashbook with the below information relating to 2/10/x9 Cashbook balance as at 1/10/x9 300 (Debit) Cash sales 200 Phone bill 30 Drawings 50 What is the closing cashbook...
-
SummerCo provides snow removal service to various clients throughout the winter. It has purchased a snow removal blade for $65,000 that it will depreciate using declining balance depreciation at 25%....
-
Business Networking solution using Windows Server 2019 /2022 with 100 to 200 employees with at least 7 departments, with security groups. Including all the servers inside Windows 2019. Setup hardware...
-
Tricare program that is a Medicare-wraparound coverage for Tricare eligible beneficiaries who have Medicare Part A and B.MIPS is the Merit-based Incentive Payment Systems that most providers will...
-
As a potential investment, you are evaluating a country/western night club called Pork Rinds and Silk Purses. You estimate a cost of $7,500,000 will be required to establish the facility. Also, you...
-
A firm offers two products for sale. The marginal cost of one product is new zero once the first unit has been produced. The marginal cost of the other product rises as output rises. What would be...
-
A rod 50 cm long and 1.0 cm in radius carries a 2.0C charge distributed uniformly over its length. Find the approximate magnitude of the electric field (a) 4.0 mm from the rod surface, not near...
-
Whats the approximate field strength 1 cm above a sheet of paper carrying uniform surface charge density s = 45 nC/m 2 ?
-
What is the electric field strength just outside the surface of a conducting sphere carrying surface charge density 1.4 C/m 2 ?
-
A point +1 C charge and another point charge Q are 40 cm apart. At the point along the line joining the two charges that is 10 cm from the +1 C charge (and hence 30 cm from charge Q), the net...
-
A point charge of +15 nC is placed at the origin of a rectangular coordinate system. a. Determine the vector electric field at P(10m, 5m, -6m). b. With the +15 nC charge still at the origin,...
-
A 1200 kg car traveling north at 10 m/s is rear-ended by a 2000 kg truck traveling at 28 m/s. What is the total momentum before and after the collision? before collision after collision kg.m/s kg.m/s

Study smarter with the SolutionInn App


