Sodium-23 (in a sample of NaCl) is subjected to neutron bombardment in a nuclear reactor to produce
Question:
Sodium-23 (in a sample of NaCl) is subjected to neutron bombardment in a nuclear reactor to produce 24Na. When removed from the reactor, the sample is radioactive, with β activity of 2.54 × 104 dpm. The decrease in radioactivity over time was studied, producing the following data: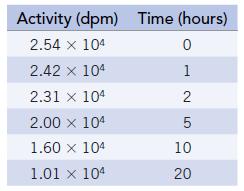
(a) Write equations for the neutron capture reaction and for the reaction in which the product of this reaction decays by β emission.
(b) Determine the half-life of sodium-24.
Transcribed Image Text:
Activity (dpm) Time (hours) 2.54 x 104 0 2.42 x 104 1 2.31 x 104 2 5 2.00 x 104 1.60 x 104 1.01 x 104 10 20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a The neutron capture reaction that produces sodium24 can be repre...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Immanuel Kant believed that the results of a decision are not as important as the reason for making it. This is the principle behind Outilitarian ethics. O deontological ethics. Rawlsian justice. O...
-
Required a. Use professional judgment in deciding on the preliminary judgment about materiality for earnings, current assets, current liabilities, and total assets. Your conclusions should be stated...
-
Data A.1 on page 136 introduces a study that examines the effect of light at night on weight gain in a sample of 27 mice observed over a fourweek period. The mice who had a light on at night gained...
-
A 0.831-g sample of SO3 is placed in a 1.00-L container and heated to 1100 K. The SO3 decomposes to SO2 and O2: At equilibrium the total pressure in the container is 1.300 atm. Find the values of Kp...
-
At a recent staff meeting, the management of Hom Technology Products was considering discontinuing the Hercules line of laptop computers from the product line. The chief financial analyst reported...
-
Who should be liable for computer viruses? Why?
-
Use a dot plot to display the data, which represent the blood glucose levels (in milligrams per deciliter) of 24 patients at a pathology laboratory. Organize the data using the indicated type of...
-
Assume that you have been hired as a consultant by CGT, a major producer of chemicals and plastics, including plastic grocery bags, styrofoam cups, and fertilizers, to estimate the firms weighted...
-
Coronado Company purchased equipment for $222,800 on October 1, 2025. It is estimated that the equipment will have a useful life of 8 years and a salvage value of $14,000. Estimated production is...
-
The average energy output of a good grade of coal is 2.6 10 7 kJ/ton. Fission of 1 mol of 235 U releases 2.1 10 10 kJ. Find the number of tons of coal needed to produce the same energy as 1 lb of...
-
A sample of wood from a Thracian chariot found in an excavation in Bulgaria has a 14 C activity of 11.2 dpm/g. Estimate the age of the chariot and the year it was made. (t for 14 C is 5.73 10 3...
-
Muniz was arrested on a charge of driving under the influence of alcohol. He was taken to a booking center, where he was asked several questions by a police officer without first being given the...
-
What, if any, advantages do you see in the corporate form over the other available alternatives (Sole proprietorship, partnership, limited liability partnership, limited partnership, limited...
-
Suppose that you are going to connect the computers in your house or apartment. What media would you use? Why? Would this change if you were building a new house?
-
Compare the corporate governance framework of the United Kingdom vis--vis India, in light of (A) corporate purpose; and (B) their law and policy-making approaches.?
-
What can traditional media do to stay relevant for the advertising world? How is traditional media going to adjust to the digital age? What might traditional media look like in the future? What are...
-
If general partners are partners who participate in management and have personal liability for the debts and obligations of the partnership, and limited partners are partners who do not participate...
-
An economist for the federal government is attempting to produce a better measure of poverty than is currently in use. To help acquire information, she recorded the annual household income (in...
-
l ask this second time correnct answer is 38,01 can we look pls Consider a non-conducting rod of length 8.8 m having a uniform charge density 4.5 nC/m. Find the electric potential at P, a...
-
Compute the time required to reduce the depth in the tank shown in Fig. 6.14 by 225 mm if the original depth is 1.38 m. The tank diameter is 1.25 m and the orifice diameter is 25 mm. dh
-
Compute the time required to reduce the depth in the tank shown in Fig. 6.14 by 1.50 m if the original depth is 2.68 m. The tank diameter is 2.25 m and the orifice diameter is 50 mm. dh
-
Compute the time required to empty the tank shown in Fig. 6.14 if the original depth is 18.5 in. The tank diameter is 22.0 in and the orifice diameter is 0.50 in. dh
-
Examine the impact of virtualization technologies, like hypervisors and containers, on resource isolation and system performance. What are the security implications of these technologies,...
-
What is the importance of the District Attorney in operating the court system? What is the single most important task solely completed by a District attorney? What are the main duties of the District...
-
9 The company has identified the following information about its overhead activity pools and the two product lines: Activity Pools Materials handling Quality control Machine maintenance Required:...

Study smarter with the SolutionInn App


