Determine whether each reaction is an oxidationreduction reaction. For each oxidationreduction reaction, identify the oxidizing agent and
Question:
Determine whether each reaction is an oxidation–reduction reaction. For each oxidation–reduction reaction, identify the oxidizing agent and the reducing agent.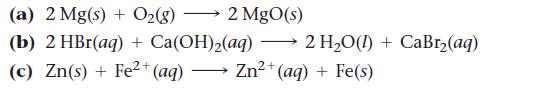
Transcribed Image Text:
(a) 2 Mg(s) + O₂(8) 2 MgO(s) (b) 2 HBr(aq) + Ca(OH)₂(aq) → 2 H₂O(I) + CaBr₂(aq) 2+ (c) Zn(s)+Fe²+ (aq) Zn²+ (aq) + Fe(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
This is a redox reaction because magnesium increases in o...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the redox reaction (a) Identify the oxidizing agent on the left side of the reaction and write a balanced oxidation half-reaction. (b) Identify the reducing agent of the left side of the...
-
(A) Is H 2 (g) an oxidizing or reducing agent in the reaction below? Explain. 2 NO 2 (g) + 7 H 2 (g) 2 NH 3 (g) + 4 H 2 O(g) (B) Identify the oxidizing agent and the reducing agent in the following...
-
Determine whether each reaction is a redox reaction. For each redox reaction, identify the oxidizing agent and the reducing agent. a. Al(s) + 3 Ag+ (aq) b. SO3(g) + HO(1) c. Ba(s) + Cl(g) d. Mg(s) +...
-
Consider an atom diffuses in a 3 D simple cubic lattice by a random walk mechanism. The atom jumps 6 x 1 0 - 5 times per second at 3 0 0 K and 3 x 1 0 4 times per second at 6 0 0 K . Assuming that...
-
Discuss the differences between traditional ERP and extended enterprise systems.
-
What is wrong with the following statement? Given any two objects, the one with the higher temperature contains more heat.
-
How do search engines make use of search? Research a few of the best-known search engines, and try to find out what kind of search algorithms they use. How efficient do you think they are at...
-
Hinshaw Company purchased a new machine on October 1, 2014, at a cost of $90,000. The company estimated that the machine has a salvage value of $8,000. The machine is expected to be used for 70,000...
-
You have a Microsoft 365 E5 subscription that contains a user named User1. You need to ensure that User1 can configure content search permission filtering. The solution must follow the principle of...
-
What is a gas-evolution reaction? Give an example.
-
Explain the principles behind an acidbase titration. What is an indicator?
-
Review the basic product concepts that underlie a successful global marketing product strategy.
-
On January 1, 2020, Everly Bottle Company sold $4,000,000 in long-term bonds. The bonds will mature in 4 years and have a stated interest rate of 10% and a yield rate of 8%. The bonds pay interest...
-
On December 31, 2020, Short Co. is in financial difficulty and cannot pay a note due that day. It is a $2,000,000 note with $200,000 accrued interest payable to Bryan, Inc. Bryan agrees to forgive...
-
How can modern computational techniques, such as molecular dynamics simulations, be used to predict phase behavior in complex mixtures, and how do these predictions inform industrial process design ?
-
Sheridan.com sells 6700 units of its product for $490 each. The selling price includes a one-year warranty on parts. It is expected that 3% of the units will be defective and that repair costs will...
-
Hurst, Inc. sold its 8% bonds with a maturity value of $9,000,000 on August 1, 2019 for $8,838,000. At the time of the sale the bonds had 5 years until they reached maturity. Interest on the bonds is...
-
Use the data from Christy Company in the chapter to answer the following: a. Calculate Christy Companys residual income for each of Year 1 through Year 5. b. Use the accounting-based equity valuation...
-
Cassandra Casey operates the Futuristic Antique Store. She maintains subsidiary ledgers for accounts payable and accounts receivable. She presents you with the following information for October 2019:...
-
You wish to replace the flooring in your kitchen with square linoleum tiles that measure 12 inches on a side. If your kitchen has a floor area of 10.7 square yards, how many such tiles will you need...
-
A typical human baby at birth is about 20 inches as measured from head to toe. An adult is much longer. What is the approximate ratio of the height of an adult female to the length of a baby at birth?
-
The solar system in perspective. The diameters of the Earth and the Sun are approximately 1.3 10 7 m and 1.4 10 9 m respectively, and the average SunEarth distance is 1.5 10 11 m. Consider a scale...
-
Data for Hermann Corporation are shown below: Selling price Variable expenses Contribution margin Percent Per Unit of Sales $ 60 39 $ 21 100% 65 35% Fixed expenses are $72,000 per month and the...
-
A Hall measurement of a semiconductor with a sheet resistance of 200 Ohm/ was performed in 0.5 Tesla, and the Hall voltage is 13 V with a Hall current of 1 A. What are the mobility and sheet carrier...
-
Cost data for Firetree Manufacturing Company for the month ended March 31 are as follows: Inventories March 1 March 31 Materials $240,000 $195,200 Work in process 428,700 510,400 Finished goods...

Study smarter with the SolutionInn App


