Determine whether each reaction is a redox reaction. For each redox reaction, identify the oxidizing agent and
Question:
Determine whether each reaction is a redox reaction. For each redox reaction, identify the oxidizing agent and the reducing agent.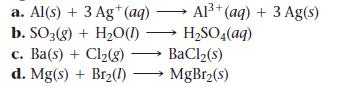
Transcribed Image Text:
a. Al(s) + 3 Ag+ (aq) b. SO3(g) + H₂O(1)→ c. Ba(s) + Cl₂(g) d. Mg(s) + Br₂(1) Al³+ (aq) + 3 Ag(s) H₂SO4(aq) → BaCl₂(s) MgBr₂(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
a Als 3 Ag aq Al3aq 3 Ags This is a redox reaction The aluminum metal ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Determine whether each reaction is a redox reaction. For each redox reaction, identify the oxidizing agent and the reducing agent. a. 4 Li(s) + O(g) 2 LiO(s) b. Mg(s) + Fe+ (aq) Mg2+(aq) + Fe(s)...
-
Consider the redox reaction (a) Identify the oxidizing agent on the left side of the reaction and write a balanced oxidation half-reaction. (b) Identify the reducing agent of the left side of the...
-
stock options are no longer as valuable and employees in startups are losing out to founders and early investors for all of these reasons except: there is higher employee turnover, less of a delay in...
-
j) Assume that one of these portfolio's is the Market Portfolio and all portfolios, except Portfolio G, are fairly priced according to the CAPM. Derive the Treynor Measure for these fairly priced...
-
The Gayton Menswear company was founded by Fred Williams in 1986 and has grown steadily over the years. Fred now has 17 stores located throughout the central and northern parts of the state. Because...
-
1. What would happen to Warner-Lambert's business if a natural disaster in Saudi Arabia depleted its natural gas resources? 2. Assess the effect on Warner-Lambert's business if the majority of...
-
The group concerned with establishing standards for flowchart symbols is ( _______________ ) (a) ASCII (b) EBCDIC (c) ANSI (d) AICPA
-
AndrewCarter, Inc. (AC), is a major Canadian producer and distributor of outdoor lighting fixtures. Its fixture is distributed throughout North America and has been in high demand for several years....
-
4. Rover on Mars. A robotic rover (initial temperature 300 K maintained by internal heating) was landed on Mars to probe the surface condition. The average surface temperature on Mars is 220 K....
-
Determine whether each redox reaction occurs spontaneously in the forward direction. a. Ni(s) + Zn+ (aq) b. Ni(s) + Pb+ (aq) c. Al(s) + 3 Ag+ (aq) d. Pb(s) + Mn+ (aq) 2+ Ni+(aq) + Zn(s) Ni+(aq) +...
-
What is the oxidation state of Cl in each ion? a. CIO- b. ClO c. ClO3 d. CIO4
-
Solve the given problems. All data are accurate to two significant digits unless greater accuracy is given The fuel for a two-cycle motorboat engine is a mixture of gasoline and oil in the ratio of...
-
Johnny and Leon are adult partners in a business selling sporting goods. The partnership records, excluding GST , for the current income year disclose the following: Receipts ($): 400,000 New sales...
-
The following budget for August, 2020 was reported for the MAINTENANCE DEPARTMENT of YESYES Manufacturing Company: YESYES MANUFACTURING COMPANY Maintenance Department Cost Budget for August 2020...
-
You are a UK bank and some time ago you agreed the interest rate and sum of a loan for forward dates from the UK's Central Bank, the Bank of England (BoE is default free). Under this agreement you...
-
A cash flow for a pass-through typically is based on some prepayment benchmark Option. Describe the benchmark Option. What does a conditional prepayment rate of 8% mean? What does 150 PSA mean? How...
-
2. What would happen if Archer-Lock and Tabaczynski did not hedge at all? No hedging means full exposure to currency risk. The company could potentially lose all of their money if USD depreciated....
-
Refer to the Applied Ecology and Environmental Research (Vol. 1, 2003) study of beech trees damaged by fungi, presented in Exercise. The researchers found that 25% of the beech trees in east central...
-
Element compound homogeneous mixture (heterogeneous mixture) 4) A piece of gold has a mass of 49.75 g. What should the volume be if it is pure gold? Gold has a density of 19.3 g/cm (3 points) D=m/v...
-
The viscosity of an oil is given as 80 SUS at 100F. Determine the viscosity in m 2 /s.
-
Convert a viscosity measurement of 6.5 10 -3 Pas into the units of lbs/ft 2 .
-
An oil container indicates that it has a viscosity of 0.12 poise at 60C. Which oil in Appendix D has a similar viscosity?
-
Consider risky Funds B and S with the following projections: Fund B S Expected return 6% 10% Standard deviation 2.5% 15% Assume the correlation between Funds B and S is -0.50. How much of your assets...
-
The Sopwith Aviation Company is considering building a new short-range commuter jet, code named 'Sky Streak!' The aircraft's engines will be powered by a new green energy source: high-tension...
-
We have two mutually exclusive projects A and B. Both require initial costs of $10,000 and last for 4 years. Project A has expected future cash flows of $4,000, $5,000, $8,000 and $3,000,...

Study smarter with the SolutionInn App


