Determine whether each redox reaction occurs spontaneously in the forward direction. a. Ni(s) + Zn+ (aq) b.
Question:
Determine whether each redox reaction occurs spontaneously in the forward direction.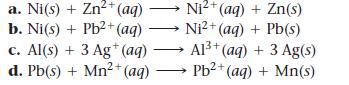
Transcribed Image Text:
a. Ni(s) + Zn²+ (aq) b. Ni(s) + Pb²+ (aq) c. Al(s) + 3 Ag+ (aq) d. Pb(s) + Mn²+ (aq) 2+ Ni²+(aq) + Zn(s) Ni²+(aq) + Pb(s) Al³+ (aq) + 3 Ag(s) Pb²+ (aq) + Mn(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
b and c oc...View the full answer

Answered By

Vijesh J
My passion to become a tutor is a lifetime milestone. Being a finance and marketing professional with hands-on experience in wealth management, portfolio management, team handling and actively contributing in promoting the company. Highly talented in managing and educating students in most attractive ways were students get involved. I will always give perfection to my works. Time is the most important for the works and I provide every answer on time without a delay. I will proofread each and every work and will deliver a with more perfection.
4.70+
5+ Reviews
15+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Determine whether each redox reaction occurs spontaneously in the forward direction. a. Ca+ (aq) + Zn(s) b. 2 Ag+ (aq) + Ni(s) c. Fe(s) + Mn+ (aq) d. 2 Al(s) + 3 Pb+ (aq) Ca(s) + Zn+ (aq) 2 Ag(s) +...
-
At 298 K, for the reaction 2 PCl 3 (g) + O 2 (g) 2 POCl 3 (l), r H = -620.2 kJ mol -1 and the standard molar entropies, in J mol 1 K 1 , are PCl 3 (g), 311.8; O 2 (g), 205.1; and POCl 3 (l), 222.4....
-
At 298 K, for the reaction 2 H + (aq) + 2 Br - (aq) + 2 NO 2 (g) Br 2 (l) + 2 HNO 2 (aq), r H = -61.6 kJ mol -1 and the standard molar entropies are H + (aq), 0 J mol -1 K -1 ; Br - (aq), 82.4 J...
-
To some U.S. manufacturers and labor unions, a cheap yuan value gives China's advantage in the global economy. bankers exports importers subsidies an unfair
-
The Cuts-n-Curves Athletic Club is a state-wide chain of full-service fitness clubs that cater to the demographics of the state (about 60% of all adults are single). The clubs each have an indoor...
-
Angie Silva has recently opened The Sandal Shop in Brisbane, Australia, a store that specializes in fashionable sandals. Angie has just received a degree in business and she is anxious to apply the...
-
Consider the following cash flow profile and assume MARR is 10 percent/year. a. Determine the ERR for this project. b. Is this project economically attractive? EOY 0 2 3 4 5 6 NCF -$100 $15 $15 $15...
-
Drew Corp. designs and manufactures mascot uniforms for high school, college, and professional sports teams. Since each team's uniform is unique in color and design, Drew uses a job order costing...
-
2 Charlie's Crispy Chicken (CCC) operates a fast-food restaurant. When accounting for its first year of business, CCC created several accounts. Account Name Description Balance Accounts Payable...
-
Suppose you wanted to cause Ni 2+ ions to come out of solution as solid Ni. Which metal could you use to accomplish this?
-
Determine whether each reaction is a redox reaction. For each redox reaction, identify the oxidizing agent and the reducing agent. a. Al(s) + 3 Ag+ (aq) b. SO3(g) + HO(1) c. Ba(s) + Cl(g) d. Mg(s) +...
-
Carrie DLake, Reed A. Green, and Doug A. Divot share a passion for golf and decide to go into the golf club manufacturing business together. On January 2, 2017,DLake, Green, and Divot form the...
-
A firm producing decorative candles lowers the price of one of its scented candles from $4 to $3.60 and finds that the weekly quantity demanded of the candles goes up from 600 per week to 630. 1....
-
Many business events are recorded in the general ledger from feeder processes. The first list below identifies eight business events. The second list specifies five feeder processes. Match the events...
-
What are the principal responsibilities, major duties, and key control concerns of each functional position pictured in Figure 8.4 on page 266 (i.e., the organization chart of an IT organization)?...
-
How do the seven control plans (five present, two missing) listed in Figure 9.6 (pg. 320) work? FIGURE 9.6 Control Matrix for Data Entry with Batches Recommended Control Plans Present Controls P-1:...
-
Suppose the economy consists of three people: Angelina, Felicia, and Marina. The table shows how their consumer spending varies as their current disposable income rises by $10,000. a. Derive each...
-
On December 1, Newton Enterprises sells $100,000 of accounts receivable to a factor, receives 85% of the value of the factored accounts, and is charged a 12% commission on the gross amount of the...
-
The Ranch 888 Noodle Company sells two types of dried noodles:ramen, at $6.50 per box, and chow fun, at $7.70 per box. So farthis year, the company has sold a total of 110,096 boxes ofnoodles,...
-
A baseball pitcher (m = 80 kg) is initially standing at rest on an extremely slippery, icy surface. He then throws a baseball (m = 0.14 kg) with a horizontal velocity of 50 m/s. What is the recoil...
-
Two cars collide head-on and lock bumpers on an icy (frictionless) road. One car has a mass of 800 kg and an initial speed of 12 m/s and is moving toward the north. The other car has a mass of 1200...
-
Two cars of equal mass are traveling as shown in Figure P7.30 just before undergoing a collision. Before the collision, one of the cars has a speed of 18 m/s along +x and the other has a speed of 25...
-
Explain what classes of stock Carter's Inc has, the characteristics of each class of stock, and the par value of the common stock. Explain how many common shares are authorized and issued and...
-
How to revised this words Typically, extended fasts are defined as fasting periods that span full days or exceed 12-16 hours?
-
how would you use/interpret the ROS and ROA ratios to analyze the company's financial statement in Albion inc?

Study smarter with the SolutionInn App


