Draw the Lewis structure for BrF with an arrow representing the dipole moment. Refer to Figure 10.10
Question:
Draw the Lewis structure for BrF with an arrow representing the dipole moment. Refer to Figure 10.10 to estimate the percent ionic character of the BrF bond.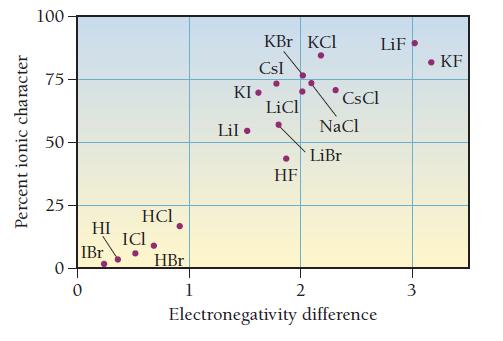
Transcribed Image Text:
Percent ionic character 100 75 50 25 0. HI IBr 0 HCI ICI. HBr 1 KBr KCI CsI KI. Lil. LiCl HF 2 CsCl NaCl LiBr LiF Electronegativity difference 3 • KF
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The Lewis structure for Brf with an arrow representing with the dipole moment and estimate the ionic ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the Lewis structure for CO with an arrow representing the dipole moment. Refer to Figure 10.10 to estimate the percent ionic character of the CO bond. Percent ionic character 100 75 50 25 0. HI...
-
The ionic character of the bond in a diatomic molecule can be estimated by the formula Where m is the experimentally measured dipole moment (in C m), e the electronic charge, and d the bond length in...
-
The Lewis structure for allene is Make a sketch of the structure of this molecule that is analogous to Figure 9.25. In addition, answer the following three questions: (a) Is the molecule planar? (b)...
-
How should employers that operate in different states and cities deal with lack of uniformity in employment laws?
-
What are database management systems? Are they the same as databases? Why are DBMSs classified as software and not hardware?
-
The financial statements for Castile Products, Inc., are given below: Account balances at the beginning of the year were: accounts receivable, $25,000; and inventory, $60,000. All sales were on...
-
Harold Fail owned a plot of land in Georgia known as Ebenezer Landing, where he operated a public boat ramp and parking lot. In 1999, Fail and Steve Bodiford entered into an oral agreement...
-
Spamela Hamderson Inc. reports the following pretax income (loss) for both financial reporting purposes and tax purposes. (Assume the carryback provision is used for a net operating loss.) The tax...
-
Consider again the market studied in Question 2, The demand curve is given by P = 100 Q and the marginal cost curve is given by MC = 10. This time we are going to assume that firms must invest funds...
-
Write the Lewis structure for each molecule or ion. a. H3COH b. OH- c. Bro- d. 0-
-
Determine if a bond between each pair of atoms would be pure covalent, polar covalent, or ionic. a. C and N b. N and S c. K and F d. N and N
-
Mandalay Industries is a private company that sells electronic test equipment. During the year 2013, the inventory records reflected the following: Units Unit Cost Total Cost Beginning...
-
Why are we interested in the sample mean and its distribution?
-
A grocery store sells an average of 478 loaves of bread each week. Sales (X) are normally distributed with a standard deviation of 17. (a) If a random sample of size n = 1 (week) is drawn, what is...
-
Briefly explain the relationship between sampling cost and sampling error. Give some examples of sampling costs.
-
The dean of student activities wants to estimate the average spending on beer per week by a student. From a previous study, the standard deviation of spending was estimated to be $39. If the dean...
-
Discuss the similarities and differences among X-charts, R-charts, S-charts, and P-charts.
-
Why might market-orientated supply-side policies have undesirable side-effects on aggregate demand?
-
A bar of length = 1 has one fixed and one free end and stiffness function c(x) = 1 - x. Find the displacement when subjected to a unit force. Pay careful attention to the boundary condition at the...
-
What is the pressure, in psig, at the bottom of a swimming pool that is 10 ft deep?
-
If air has a constant specific weight of 0.075 lb/ft 3 , what pressure difference would result when driving from the base to the top of Pikes Peak, if the climb for the trip is 8400 ft?
-
Figure 4.21 shows a vacuum tank with a flat circular observation window in one end. If the pressure in the tank is 0.12 psia when the barometer reads 30.5 in of mercury, calculate the total force on...
-
For some scalar c ER, What is T(ca)? (c). Find a linear transformation T: R2 R2 such that T(1,2) = (3,0) > and T(2,1)=(1,2). Solution Express your answer in the form : T(x, y): -x+ay cx+dy = where a...
-
Solve the following equation. X 1 3 X + 6 4
-
Canada 13. How much less was the fraction OF Sales United Fraction of Total Sales, 2000 For England States than For the United States? Australia 10 20 718 Japan 15 911 1T England

Study smarter with the SolutionInn App


