Draw the Lewis structure (including resonance structures) for methyl azide (CH 3 N 3 ). For each
Question:
Draw the Lewis structure (including resonance structures) for methyl azide (CH3N3). For each resonance structure, assign formal charges to all atoms that have formal charge.

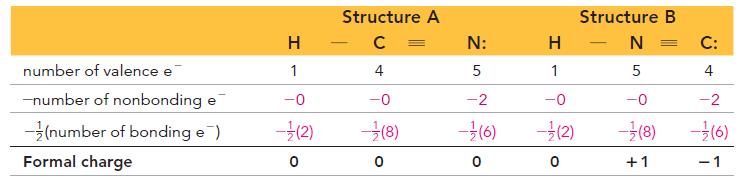
Transcribed Image Text:
Formal charge number of valence electrons - (number of nonbonding electrons + number of bonding electrons) So the formal charge of hydrogen in HF is 0: Formal charge = 1- [0+ (2)]=0 Number of valence electrons for H Number of electrons that H "owns" in the Lewis structure Similarly, we calculate the formal charge of fluorine in HF as 0: Formal charge = 7 - [6+ (2)] = 0 Number of valence electrons for F Number of electrons that F "owns" in the Lewis structure
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (6 reviews)
H HCNNN H H HCNNN H I II For structureI Formal charge for all the three Hatom...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the Lewis structure (including resonance structures) for the acetate ion (CH 3 COO ). For each resonance structure, assign formal charges to all atoms that have formal charge. Formal charge =...
-
Draw the Lewis structure (including resonance structures) formethyl azide (CH3N3). For each resonance structure, assign formalcharges to all atoms that have formal charge. Draw the Lewis dotstructure...
-
Draw the Lewis structure (including resonance structures) for nitromethane (CH 3 NO 2 ). For each resonance structure, assign formal charges to all atoms that have formal charge.
-
Problem 3: Dorado and Maya are the children of Jess and Sabel. In November 2000 Sabel died intestate leaving P2,000,000 estate before P1,000,000 deductions and P15,000 estate tax. How would the...
-
What are data warehouses? How are they like databases? How do they differ from databases?
-
Abdou Co. produces advertising videos. During the last six months of the current fiscal year, Abdou Co. received the following notes: Instructions 1. Determine for each note (a) The due date and (b)...
-
Nick Spanos Antiques issued its \(8 \%, 10\)-year bonds payable at a price of \(\$ 440,000\) (maturity value is \(\$ 500,000\) ). The company uses the straight-line amortization method for the bonds....
-
Suppose that during 2013, the market for Stanmore?s special-purpose machines grew by 3%. In Exercise 12- 22, Stanmore Corporation makes a special- purpose machine, D4H, used in the textile industry....
-
How do small restaurants that have challenges competing with larger chains that have more visibility and money to spend on advertising? Social media is suggested as a way to raise awareness of...
-
What are the formal charges of the atoms shown in red? CH3 CH3N0: -N- CH3
-
How important is the resonance structure shown here to the overall structure of carbon dioxide? Explain. :0=C:
-
Describe the effects of rental ceilings. Who benefits from rental ceilings? Who suffers? What are the long- term effects of rental ceilings? How can landlords and tenants cheat on rental ceilings? Do...
-
Critically explain the target group of the given program in the following topics based on the evidence derived from the given program. Use credible evidence to support your point of view Explain the...
-
Identify one type of business and describe that business and it's lending needs. Then describe what their potential lending needs may? Manufacturing Retail Wholesale Service Construction
-
The risk-free interest rate is 3% annualized and continuously compounded. Assume the market is frictionless. When you plot the forward curves below, you may choose to either (i) use the analytical...
-
The SickKids Foundation is the largest charitable funder of child health research, learning, and care in Canada. They have been successful in raising funds to support the Hospital for Sick Children...
-
Calculate the alpha and beta of Stock A 2) Please calculate the volatility of the portfolio (stock a and stock b) based on two scenarios (a. equal weight b. arbitrary weight: stock a 20% stock b 80%...
-
Projected unit sales for Astor Corp. for the last half of 2009 are Finished goods inventory on June 30, 2009 is 7,400 units. The company tries to keep an ending inventory of 25 percent of the...
-
The Alert Company is a closely held investment-services group that has been very successful over the past five years, consistently providing most members of the top management group with 50% bonuses....
-
For the manometer shown in Fig. 3.30, calculate (p A - p B ). Water IB 150 mm - Mercury (sg = 13.54) 900 mm Oil (sg = 0.86) 600 mm
-
For the compound manometer shown in Fig. 3.31, calculate the pressure at point A. Oil (sg = 0.90) Water 125 mm 475 mm 250 mm 50 mm Mercury (sg = 13.54)
-
For the compound differential manometer in Fig. 3.32, calculate (p A - p B ). Water, Oil (sg = 0.90) 6 in 8 in 6 in 10 in 6 in Mercury (sg = 13.54)
-
Place the following phasesa job moves through in order of processing: - 1. 2. 3. 4. 5. 6. Purge - 1. 2. 3. 4. 5. 6. Conversion - 1. 2. 3. 4. 5. 6. Input - 1. 2. 3. 4. 5. 6. Output - 1. 2. 3. 4. 5. 6....
-
Identify and describe different medical claims created byhealthcareproviders that are submitted to insurance companies for payment. Why is it important for healthcare administrators to be very...
-
Write a JCL Data Definition statement with the following characteristics. 1) The ddname is INPUT1. 2) The dataset name is KC03D11.TEST2.DATA. 3) You can share the dataset with other users. Notes: 1)...

Study smarter with the SolutionInn App


