What are the formal charges of the atoms shown in red? CH3 CH3N0: -N- CH3
Question:
What are the formal charges of the atoms shown in red?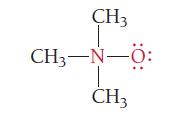
Transcribed Image Text:
CH3 CH3−N–0: -N- CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
N has a fo...View the full answer

Answered By

Shaira grace
I have experience of more than ten years in handing academic tasks and assisting students to handle academic challenges. My level of education and expertise allows me communicate eloquently with clients and therefore understanding their nature and solving it successfully.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What are the formal charges of the atoms shown in red? :: CH3-S-CH3
-
In the Lewis structure shown here, A, D, E, Q, X, and Z represent elements in the first two rows of the periodic table (H--- Ne). Identify all six elements so that the formal charges of all atoms are...
-
Consider the following separate cases pertaining to Audit Opinions: (a) You have audited Froggy No 1 Ltd. During the audit you discover that a material amount of inventory is held in a robotic...
-
Scandinavo Ltd. is a CCPC that began operations on January 1, 2020 when it was first incorporated and a calendar fiscal period was chosen. Scandinavo Ltd. Is not associated with any other...
-
Why would a company be interested in creating a data warehouse? Why would a company not be interested in creating a data warehouse?
-
Paul Swanson has an opportunity to acquire a franchise from The Yogurt Place, Inc., to dispense frozen yogurt products under The Yogurt Place name. Mr. Swanson has assembled the following information...
-
Zachary and Carrie Sexton (the Buyers) were searching for a home in the Kings wood neighborhood of Atlanta, Georgia. The Buyers real estate agent learned that Russell and Linda Sewell (the Sellers)...
-
In 2013, the Westgate Construction Company entered into a contract to construct a road for Santa Clara County for $10,000,000. The road was completed in 2015. Information related to the contract is...
-
Discuss how a good understanding of the state of the economy might help you make better decisions. Discuss how much of a role you think a government should play in a market system. Explain how you...
-
Write the Lewis structure for each molecule (octet rule not followed). a. BCl 3 b. NO 2 c. BH 3
-
Draw the Lewis structure (including resonance structures) for methyl azide (CH 3 N 3 ). For each resonance structure, assign formal charges to all atoms that have formal charge. Formal charge number...
-
In how many ways can 3 people be seated in a circle? 4? n?
-
How do macroeconomists distinguish between flexible and sticky prices and wages?
-
Discuss the validity of the following statement: Unlike Solows model, Romers model concludes that changes in the saving rate do not affect the sustained per-capita output growth rate.
-
Discuss the following statement: When Keynes stated that in the long run, we are all dead he meant that we should focus only on the short run and not pay attention to any long-run consequences of our...
-
Use the following table to find the steady-state values of the capital-labor ratio and output per worker (i.e., complete the table) if the per worker production function is yt = 2k t 0.3 : S Saving...
-
Distinguish between procyclical and countercyclical economic variables.
-
Compare the relative effectiveness of fiscal and monetary policy under (a) fixed; (b) free-floating exchange rates. How is the effectiveness influenced by the elasticity of supply of international...
-
Calculate the number of neutrons of 239Pu.
-
For the differential manometer shown in Fig. 3.27, calculate the pressure difference between points A and B. The specific gravity of the oil is 0.85. 10 in 32 in Oil Water 9 in el
-
For the manometer shown in Fig. 3.28, calculate (p A - p B ). Oil (sg = 0.85) 8 in Water 33 in 12 in
-
For the manometer shown in Fig. 3.29, calculate (p A - p B ). Oil l 150 mm (sg = 0.90) Water 750 mm Mercury (sg = 13.54) 500 mm
-
photography business Is personal selling appropriate for your product, target market, and marketing environment? Take time to consider whether you need your own sales force or whether you can train...
-
How might globalization be a problem for a successful national company that is intent on going international? What advantages would the national company have by going international? Provide an...
-
The Clorox Company: Leveraging Green for Growth Briefly outline a marketing plan for each of the three sustainable brands going forward. Which of them would you be most optimistic about? Why? What...

Study smarter with the SolutionInn App


