For each pair of substances, choose the one that you expect to have the higher standard molar
Question:
For each pair of substances, choose the one that you expect to have the higher standard molar entropy (S°) at 25 °C. Explain your choices.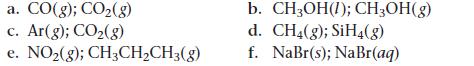
Transcribed Image Text:
a. CO(g); CO₂(g) c. Ar(g); CO₂(g) e. NO₂(g); CH3CH₂CH3(g) b. CH₂OH(1); CH3OH(g) d. CH₂(g); SiH4(g) f. NaBr(s); NaBr(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a CO 2 g greater molar mass and comple...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each pair of substances, choose the one that you expect to have the higher standard molar entropy (S) at 25 C. Explain your choices. a. NaNO3(s); NaNO3(aq) c. Br(1); Br(g) e. PC13(g); PC15(g) b....
-
For each pair of substances listed here, choose the one having the larger standard entropy value at 25C. The same molar amount is used in the comparison. Explain the basis for your choice. (a) Li(s)...
-
For each pair of firms, choose the one that you think would be more sensitive to the business cycle. a. General Autos or General Pharmaceuticals. b. Friendly Airlines or Happy Cinemas.
-
Under what circumstance is it most appropriate to travel in reverse with a load? A. When you are carrying a non-standard load B. When a tall load obstructs your forward vision C. When the load is...
-
What are the objectives of economically developing countries? How do these objectives relate to marketing? Comment.
-
Bronkowski is a retailer for high-tech recording disks. The projected operating profit for the current year is $200,000 based on a sales volume of 200,000 units. The company has been selling the...
-
On April 23, 2014, Calvin Loyer admitted his wife, Edeltrud Loyer, to a nursing home administered by Signature Healthcare. During the admissions process, Calvin signed an arbitration agreement...
-
Brian Smith, network administrator at Advanced Energy Technology (AET), was given the responsibility of implementing the migration of a large data center to a new office location. Careful planning...
-
1. Suppose that x1,. ,...,xk+1 is a circuit of length k in a graph G (x = xk+1) of length 3 or greater (k 3). Suppose as well that the circuit does not repeat edges (ie xx+1 xjxj+1 for i j). Show...
-
What is the molar entropy of a pure crystal at 0 K? What is the significance of the answer to this question?
-
How does the molar entropy of a substance change with increasing temperature?
-
Suppose that the following table is your record of exam grades in your Principles of Economics course: Use these data to make up a table of total, average, and marginal grades for the five exams....
-
Why is promoting responsible gaming important for MGM? Why is MGM interested in addressing this issue?
-
A protein molecule of human serum albumin in a physiological solution undergoes a denaturation transition with the increase in temperature. The transition happens around 70 degrees Celsius in a...
-
Explain three (3) ways the teacher assistants used daily interaction with students to demonstrate the different styles and functions of verbal communication. For each strategy used, Explain what...
-
Why did the Hawthorne studies and McGregor's Theory X and Theory Y have an impact on the study of organizational behavior? 2. Explain why and how changes in the U.S. demographics affect the...
-
Briefly explain the current procedures and process for a cruise passenger boarding/exiting a ship in Miami, Florida. Passenger is departing Toronto, Canada on the same day as the cruise departs Port...
-
In 1954, Brown v. Board of Education desegregated schools in the United States and deemed desegregated schools separate but unequal. Do you think the Supreme Court decision was an appropriate...
-
Banner Company acquires an 80% interest in Roller Company for $640,000 cash on January 1, 2013. The NCI has a fair value of $160,000. Any excess of cost over book value is attributed to goodwill. To...
-
A car starts from rest and then accelerates uniformly to a linear speed of 15 m/s in 40 s. If the tires have a radius of 25 cm, what are the magnitudes of (a) The average linear acceleration of a...
-
A car is traveling with a speed of 20 m/s. If the tires have an angular speed of 62 rad/s, what is the radius of the tires?
-
Consider a tennis ball that is hit by a player at the baseline with a horizontal velocity of 45 m/s (about 100 mi/h). The ball travels as a projectile to the players opponent on the opposite...
-
Describe the entrepreneurial characteristics of Amazon. How has Bezos promoted innovation within the organization? Explain.
-
Please view the image for Activity #1 . Then describe a purchase scenario (real or imagined) using the Consumer Decision Process. Also, describe the role of at least three external and internal...
-
1. A Farm is producing groundnuts and soya bean on 100 acres of land and 16 goats on 2 hectares of land. Suppose groundnuts occupies 65 percent and soya bean 35 percent of the total crop area. Yield...

Study smarter with the SolutionInn App


