For each reaction, calculate H rxn , S rxn , and G rxn at 25 C and
Question:
For each reaction, calculate ΔH°rxn, ΔS°rxn, and ΔG°rxn at 25 °C and state whether or not the reaction is spontaneous. If the reaction is not spontaneous, would a change in temperature make it spontaneous? If so, should the temperature be raised or lowered from 25 °C?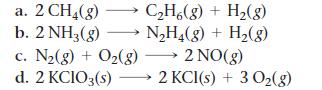
Transcribed Image Text:
a. 2 CH₂(g) b. 2 NH3(g) C₂H6(g) + H₂(g) N₂H₂(g) + H₂(g) c. N₂(g) + O₂(g) →→→ 2 NO(g) d. 2 KCIO3(s)→→→→→2 KCl(s) + 3 O₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
To calculate Hrxn Srxn and Grxn we need the standard enthalpy of formation Hf values and standard en...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Suppose that your company has decided to invest RM316 million in a 5-year construction project. The project is expected to be sold for RM200 million once completed. Estimate the Internal Rate of...
-
Use standard free energies of formation to calculate G at 25 C for each reaction in Problem 62. How well do the values of G calculated this way compare to those calculated from H and S? Which of the...
-
Use standard free energies of formation to calculate G at 25 C for each reaction in Problem 61. How do the values of G calculated this way compare to those calculated from H and S? Which of the two...
-
Why is it important to have a defined project scope? Why is it important to make sure there is agreement about the scope? Is there anything in the "Why Should You Use the WBS?
-
Discuss the distinguishing features of the Japanese distribution system.
-
Late one night on a highway, a car speeds by you and fades into the distance. Under these conditions, the pupils of your eyes have diameters of about 7.0 mm. The taillights of this car are separated...
-
Suppose you started up your own landscaping business. A customer paid you $120 in advance to mow his or her lawn while he or she was on vacation. You performed landscaping services for a local...
-
What is the difference between the conventional test data approach and the integrated test facility approach?
-
Delta Company produces a single product. The cost of producing and selling a single unit of this product at the company's normal activity level of 105,600 units per year is: Direct materials Direct...
-
Determine G for the reaction: Use the following reactions with known G rxn values: FeO3(s) + 3 CO(g) 2 Fe(s) + 3 CO(g)
-
In photosynthesis, plants form glucose (C 6 H 12 O 6 ) and oxygen from carbon dioxide and water. Write a balanced equation for photosynthesis and calculate H rxn , S rxn , and G rxn at 25 C. Is...
-
Find the trigonometric Fourier series for the waveform shown.
-
The distance between the planes of a crystal is sought using a diffractometer. X-rays with a wavelength of 0.135 nm are used, and the angle for the first order diffraction is 0 9.94. What is the...
-
Question 1) Identify how evolution of IT systems affects organization design and inter-organizational relationships. Question 2) Describe how IT is used for decision making and control of the...
-
McGill and Smyth have capital balances on January 1 of $41,000 and $34,000, respectively. The partnership income-sharing agreement provides for (1) annual salaries of $25,000 for McGill and $17,000...
-
Given the following C++ source code: #include using namespace std; class Residential Area { wwwww private: public: int house; //number of houses int resident; //number of residents void setData (h,...
-
The defense of necessity occurs when an individual has a need to break the law in order to prevent serious harm (Matthew Lippman, 2021). As an example, if a person encounters two people engaged in a...
-
On January 1, 2017, Evers Company purchased the following two machines for use in its production process. Machine A: The cash price of this machine was $48,000. Related expenditures included: sales...
-
Which, if any, of the dichloroethene molecules drawn in Data Table II (3.) (4.) and (5.) are geometric isomers? A. B. C. D. cis-1,2-dichloroethene and trans-1,2-dichloroethene...
-
Consider the flagpole in Figure P8.29. If the flagpole has a mass of 20 kg and length 10 m and the angle the cable makes with the pole is ?? = 25, what are the magnitude and direction of the force...
-
A painter is standing on the ladder (mass 40 kg and length 2.5 m) in Figure P8.30. There is friction between the bottom of the ladder and the floor with S = 0.30, but there is no friction between...
-
Consider again the ladder in Problem 30. What is the sign of the torque on the ladder due to the force from the wall? Data from Problem 30 A painter is standing on the ladder (mass 40 kg and length...
-
How do advanced thermodynamic models, such as the Wilson equation or NRTL (Non-Random Two-Liquid) model, assist in predicting and analyzing the absorption behavior in multi-component systems ?
-
Discuss the role of mass transfer coefficients and the impact of interfacial phenomena in gas-liquid absorption processes. How do hydrodynamic conditions influence the efficiency of mass transfer ?
-
Please provide feedback and improve this argument for the topic the offence/defence of infanticide ultimately just an objectionable patriarchal condescension toward women. Disregards socio-political...

Study smarter with the SolutionInn App


