Gallium has two naturally occurring isotopes with the following masses and natural abundances: Sketch the mass spectrum
Question:
Gallium has two naturally occurring isotopes with the following masses and natural abundances: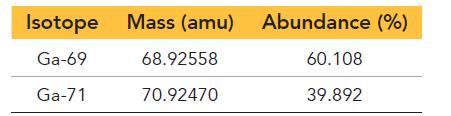
Sketch the mass spectrum of gallium.
Transcribed Image Text:
Isotope Ga-69 Ga-71 Mass (amu) Abundance (%) 60.108 39.892 68.92558 70.92470
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Fig Mass spec...View the full answer

Answered By

Daniel Kimutai
I am a competent academic expert who delivers excellent writing content from various subjects that pertain to academics. It includes Electronics engineering, History, Economics, Government, Management, IT, Religion, English, Psychology, Sociology, among others. By using Grammarly and Turnitin tools, I make sure that the writing content is original and delivered in time. For seven years, I have worked as a freelance writer, and many scholars have achieved their career dreams through my assistance.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Patients seeking care at the County General emergency room wait, on average, 6 minutes before seeing the triage nurse who spends, on average, 4 minutes assessing the severity of their problem. The...
-
Magnesium has three naturally occurring isotopes with the following masses and natural abundances: Sketch the mass spectrum of magnesium. Isotope Mg-24 Mg-25 Mg-26 Mass (amu) 23.9850 24.9858 25.9826...
-
An element has two naturally occurring isotopes with the following masses and abundances: Isotopic Mass (amu) Fractional Abundance 49.9472. 2.500 103 50.9440. 0.9975 What is the atomic mass of this...
-
Draw structures for the following molecules (a) Acrylonitrile, C3H3N, which contains a carbon-carbon double bond and a carbon-nitrogen triple bond (b) Ethyl methyl ether, C3H8O, which contains an...
-
Rainbow Tours gives walking tours of Springfield. Rainbow charges $40 per person for the tour and incurs $16 in variable costs for labor, drinks, and maps. The monthly fixed costs for Rainbow Tours...
-
The Smart Way Products Company manufactures and sells wireless video cell phone, which it guarantees for five years. If a cell phone fails, it is replaced free, but the customer is charged a service...
-
Pelligrini Electronics, Corp., was recently formed as a corporation. The balance of each item in the companys accounting equation is shown for November 1 and for each of the following business days:...
-
Amy and Lester are partners in operating a store. Without consulting Amy, Lester enters into a contract to purchase merchandise for the store. Amy contends that she did not authorize the order and...
-
Crane Industries incurs unit costs of $6 ($4 variable and $2 fixed) in making an assembly part for its finished product. A supplier offers to make 13,500 of the assembly part at $5 per unit. If the...
-
Carl arrives at Dewey, Dewey, and Howe. He is told he will be working with Attorney Howe. Carl meets with Attorney Howe, and they discuss a number of matters, including the flat fees Howe charges for...
-
Which pair of elements do you expect to be most similar? Why? a. Nitrogen and oxygen b. Titanium and gallium c. Lithium and sodium d. Germanium and arsenic e. Argon and bromine
-
Which pair of elements do you expect to be most similar? Why? a. N and Ni b. Mo and Sn c. Na and Mg d. Cl and F e. Si and P
-
Extend the heap abstraction with three operations: allocArray, getAt, and setAt, which allows for treating a chunk of memory as a two-dimensional array. Given the row and column sizes, allocArray...
-
Explain why engagements to perform agreed-upon procedures are restricted to specified users.
-
Contrast a WebTrust engagement with a SysTrust engagement in terms of the subject matter and the intended users.
-
a. What representations are made by the auditor in the auditor's standard report? b. What circumstances justify the issuance of an unqualified opinion?
-
Describe the four principles of a reliable system included in the criteria used for a SysTrust engagement.
-
a. What are the essential characteristics of a profession? b. Give two examples of each characteristic that enables public accounting to be a profession.
-
The following data represent the accounting results for the year ended December 31, 2013, for four different manufacturing corporations. The effective tax rates were as follows: 30% for 2011, 32% for...
-
A city maintains a solid waste landfill that was 12 percent filled at the end of Year 1 and 26 percent filled at the end of Year 2. During those periods, the government estimated that total closure...
-
There are two different compounds with molecular formula C 2 H 6 O. One of these isomers has a much higher boiling point than the other. Explain why.
-
Determine the relationship between the two structures below. Are they resonance structures or are they constitutional isomers?
-
In each reaction, identify the Lewis acid and the Lewis base: (a) (b) (c) F L-
-
Through play, children refine their movements to achieve their goals. At all levels of development, you can find similar examples of how play fosters learning for a child. The concept and value of...
-
Insert Design Layout References Mailings Review View Help Acrobat st_Practice Template.v1.0 Saved to this PC - Tell me what you want to do AE.Attie Sh H 10 11 121 18 14 15 16 17 18 N 20.1 21. 22 123...
-
Your supervisor is planning a trip to China to discuss a joint venture to get your company's product into the Chinese market. Provide your supervisor with information about cultural differences to be...

Study smarter with the SolutionInn App


