Given a barometric pressure of 762.4 mmHg, calculate the pressure of each gas sample as indicated by
Question:
Given a barometric pressure of 762.4 mmHg, calculate the pressure of each gas sample as indicated by the manometer.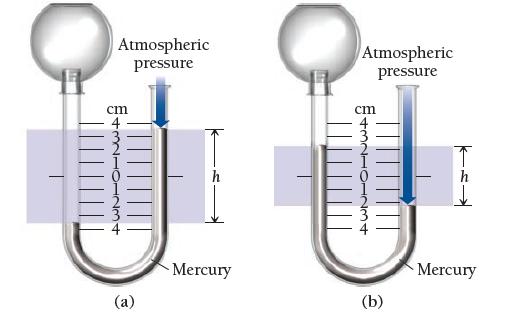
Transcribed Image Text:
Atmospheric pressure cm (a) Mercury Atmospheric pressure cm 11 (b) Mercury
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a 83...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Given a barometric pressure of 751.5 mmHg, calculate the pressure of each gas sample as indicated by the manometer. Atmospheric pressure cm (a) I Mercury Atmospheric pressure cm 4 MNOHY+ (b) Mercury
-
An 89.3 mL sample of wet O 2 (g) is collected over water at 21.3 C at a barometric pressure of 756 mmHg (vapor pressure of water at 21.3 C = 19 mmHg). (a) What is the partial pressure of O 2 (g) in...
-
(A) The reaction of aluminum with hydrochloric acid produces hydrogen gas. The balanced chemical equation for the reaction is given below. 2 Al(s) + 6 HCl(aq) 2 AlCl 3 (aq) + 3 H 2 (g) If 35.5 mL of...
-
How does the ERM system help with timely, effective, and democratic business decisions? A) Reporting to the presidents of each unit a faculty in a method that allows silo type decisions B) Board...
-
What is an enterprise system supposed to accomplish?
-
A rectangular plate has a length of (21.3 0.2) cm and a width of (9.8 0.1) cm. Calculate the area of the plate, including its uncertainty.
-
a. What is a proof of cash? b. What type of test is a proof of cash? c. How should a proof of cash be prepared? d. Explain the treatment of reconciling items in a proof of cash.
-
Chris Lane, the head professional at Royal Oak Country Club, must develop a schedule of matches for the couples golf league that begins its season at 4:00 P.M. tomorrow. Eighteen couples signed up...
-
Designing the logic for a program that calculates a person's BMI. We are using Flowgorithm for this project. It is supposed to loop continuously until the user enters 0 for the height in inches. The...
-
A sample of gas has an initial volume of 5.6 L at a pressure of 735 mmHg. If the volume of the gas is increased to 9.4 L, what is its pressure?
-
The North American record for highest recorded barometric pressure is 31.85 in Hg, set in 1989 in Northway, Alaska. Convert this pressure to each indicated unit. a. MmHg b. Atm c. Torr d. KPa...
-
Making use of the fact that the Helmholtz free energy \(A(N, V, T)\) of a thermodynamic system is an extensive property of the system, show that \[ N\left(\frac{\partial A}{\partial N}ight)_{V,...
-
On 15 November 19X8, Hughes consigned 300 cases of wooden items to Galvez of Madrid. On 31 December 19X8, Galvez forwarded an account sales, with a draft for the balance, showing the following...
-
A 7 4 0 0 kg rocket blasts off vertically from the launch pad with a constant upward acceleration of 2 . 2 0 m / s 2 and feels no appreciable air resistance. When it has reached a height of 6 0 0 m ,...
-
Epson produces color cartridges for inkjet printers. Suppose cartridges are sold to mail-order distributors for $5.20 each. Total fixed costs per year are $820,800. Variable cost per unit are $1.85...
-
An object moves in the positive x - direction with a constant acceleration. At x = 4 . 9 0 m , its speed is 7 . 0 0 m / s; 2 . 2 0 s later, the object is at x = 6 0 . 0 m . What is the magnitude of...
-
A company has fixed costs of $104,000 and it can produce 920 items for a total cost of $108,600. Find the linear cost function, C (x)
-
The WorldCom fraud described in the chapter involved a simple earnings manipulation through which the company capitalized expenses to increase net income. This transaction was recorded by a journal...
-
The overall reaction and equilibrium constant value for a hydrogenoxygen fuel cell at 298 K is 2H 2 (g) + O 2 (g) 2H 2 O(l) K = 1.28 10 83 a. Calculate E cell and G 8 at 298 K for the fuel cell...
-
Determine the vertical displacement of joint D. Using Castiglianos theorem. AE is constant. Assume the members are pin connected at their ends. 000 4 m 4 m - 15 kN 20 kN 000
-
Determine the vertical displacement of joint D. Use the method of virtual work. AE is constant. Assume the members are pin connected at their ends. 000 4 m 4 m - 15 kN 20 kN 000
-
Determine the vertical displacement of joint E. For each member A = 400 mm 2 , E = 200 GPa. Using Castiglianos theorem. 1.5 m A JO 45 kN 2 m 2 m
-
Revenues $ 5 , 0 0 0 , 0 0 0 COGS $ 2 , 0 0 0 , 0 0 0 Gross Profits $ 3 , 0 0 0 , 0 0 0 Depreciation $ 5 0 0 , 0 0 0 EBIT $ 2 , 5 0 0 , 0 0 0 Interest $ 5 0 0 , 0 0 0 EBT $ 2 , 0 0 0 , 0 0 0 Taxes $...
-
Solve for x. -7+ log, (x+3)=-5
-
Fooddservice management: Role Playing Scenario below: The Busy Preceptor! You're just started your food systems experience as a student at a Hospital's Food & Nutrition Service cafeteria and patient...

Study smarter with the SolutionInn App


