How many types of hybrid orbitals do we use to describe each molecule? a. NO5 b. CH5NO
Question:
How many types of hybrid orbitals do we use to describe each molecule?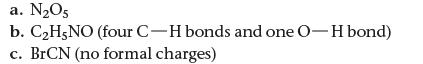
Transcribed Image Text:
a. N₂O5 b. C₂H5NO (four C-H bonds and one O-H bond) c. BrCN (no formal charges)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
a ...View the full answer

Answered By

Rinki Devi
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions.
Hi there! Are you looking for a committed, reliable, and enthusiastic tutor? Well, teaching and learning are more of a second nature to me, having been raised by parents who are both teachers. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students.
I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and helped them achieve great subject knowledge.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Describe the various types of hybrid orbitals and how they are used to describe the bonding in alkanes, alkenes, and alkynes. How does hybridization explain that in allene, CH 2 =C=CH 2 , the two CH...
-
An atom in a molecule has two bonds to other atoms and one lone pair. What kind of hybrid orbitals do you expect for this atom? Describe how you arrived at your answer.
-
A school computer technician who was remotely accessing the hard drives of school-issued laptops for routine maintenance found sexually explicit images of a Grade 10 student on a high school...
-
Jeremiah Wedgewood, the CFO, is adamant that the company needs to move ahead with the new division. While Josey also thinks that creating a new division with a new product line is a good idea, she is...
-
Renfree Mines, Inc., owns the mining rights to a large tract of land in a mountainous area. The tract contains a mineral deposit that the company believes might be commercially attractive to mine and...
-
Was John a good team member? Was he a responsible team member? Explain.
-
How to prepare a witness for deposition?
-
What is the value of a preferred stock where the dividend rate is 14 percent on a $100 par value, and the markets required yield on similar shares is 12 percent?
-
Please solve correctly Explain the basic differences between. the Marketing Mix described by Kotler in his book and the Film Marketing Mix described by Finola Keriggan, Explain the meaning of the two...
-
The most stable forms of the nonmetals in groups 4A, 5A, and 6A of the second period are molecules with multiple bonds. Beginning with the third period, the most stable forms of the nonmetals of...
-
The results of a molecular orbital calculation for NH 3 are shown here. Examine each of the orbitals and classify them as bonding, antibonding, or nonbonding. Assign the correct number of electrons...
-
A manager can invoke the "employment-at-will" doctrine and fire a non-union worker who complains about too much overtime.
-
Tytos borrowed $16,500 to pay for the wedding of his daughter, Genna, to Emmon Frey. Tytos repaid the loan after 3 years with a single payment of $19,224. Assuming the interest compounded monthly,...
-
A proposed new investment has projected sales of $515,000. Variable costs are 40 percent of sales, and fixed costs are $134,500; depreciation is $52,750. Prepare a pro forma income statement assuming...
-
A particular bond pays $4580 at the end of 1 year, $3270 at the end of 2 years and $5640 at the end of 3 years. The yield to maturity for the bond is 0.11. What is the present value of this bond?...
-
Suppose a 0 . 3 6 6 kg object is sliding on a frictional surface with k of 0 . 0 9 0 . What is the magnitude of the force of friction in N ?
-
Contributions to a spouse's RRSP [cause / do not cause] attribution. (This is roughly true - as long as they money remains within the RRSP for a few years.) This uses up [your / your spouse's] RRSP...
-
Compare and contrast the two common-size balance sheets below. Which one do you think may belong to an auto manufacturer? To a computer manufacturer?
-
a. Determine the domain and range of the following functions.b. Graph each function using a graphing utility. Be sure to experiment with the window and orientation to give the best perspective of the...
-
The 2014-T6 aluminum rod has a diameter of 0.5 in. and is lightly attached to the rigid supports at A and B when T 1 = 70°F. Determine the force P that must be applied to the collar so that, when...
-
The 2014-T6 aluminum rod has a diameter of 0.5 in. and is lightly attached to the rigid supports at A and B when T 1 = 70°F. If the temperature becomes T 2 = 10°F, and an axial force of P =...
-
The force P is applied to the bar, which is made from an elastic perfectly plastic material. Construct a graph to show how the force in each section AB and BC (vertical axis) varies as P (horizontal...
-
How do team leadership dynamics, including emergent and appointed leadership roles, influence team effectiveness and member satisfaction, and what leadership approaches are most effective in...
-
If you were to evaluate lim 2 f(x) using the Table Method, which formula(s) defining f(x) would be used to generate table values?
-
what ways do emergent social structures, such as informal roles, networks, and cliques, influence team cohesion and information flow, and how can organizations leverage these structures to facilitate...

Study smarter with the SolutionInn App


