Identify the correct balanced equation for the combustion of propane (C 3 H 8 ). a) C3H8(g)
Question:
Identify the correct balanced equation for the combustion of propane (C3H8).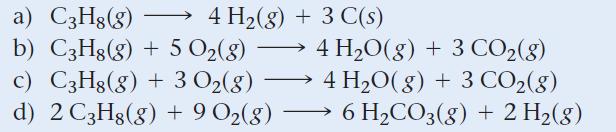
Transcribed Image Text:
a) C3H8(g) b) C3H8(g) + 5 0₂(8) c) C3H8(g) + 3 O₂(g) d) 2 C3H8(g) + 9 0₂(8) → 4 H₂(g) + 3 C(s) — - 4 H₂O(g) + 3 CO₂(g) → 4 H₂O(g) + 3 CO₂(g) 6 H₂CO3(g) + 2 H₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
b C3H...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
QUESTION 1 When propane undergoes complete combustion, the products are carbon dioxide and water.? ? ? ? __ C 3 H 8 (g) + __ O 2 (g) ? __ CO 2 (g) + __ H 2 O(g)What are the respective coefficients...
-
Propane C3H8 is burned with air, and the combustion products consist of CO2, CO, H2O, O2, N2, OH, H2, and NO. The number of equilibrium constant relations needed to determine the equilibrium...
-
A gasoline engine is converted to run on propane as shown in Fig. P13.144. Assume the propane enters the engine at 25C, at the rate of 40 kg/h. Only 90% theoretical air enters at 25C, so 90% of the C...
-
One item is omitted in each of the following summaries of balance sheet and income statement data for the following four different corporations: Determine the missing amounts, identifying them...
-
Zevon, Inc., has 9 percent coupon bonds on the market that have 8 years left to maturity. The bonds make annual payments. If the YTM on these bonds is 7 percent, what is the current bond price?
-
A circular grill of diameter 0.25 m and emissivity 0.9 is maintained at a constant surface temperature of 130C. What electrical power is required when the room air and surroundings are at 24C?
-
In 2014, Air Asia Flight No. 8501 crashed in the Java Sea while flying from Indonesia to Singapore. The crash resulted in the deaths of all passengers and crew on board. The plane involved in the...
-
Assume that you are the president of Highlight Construction Company. At the end of the first year (December 31, 2014) of operations, the following financial data for the company are available:...
-
You are given the following algorithm written in pseudocode where the entries of the input array A and the input k are positive integer numbers. Also, assume that array A has sufficiently large...
-
You just began a position as a financial accountant at Peyton Approved. In this role, your first task is to prepare the companys financials for the year-end audit. Additionally, the company is...
-
Write a balanced equation for the combustion of liquid methyl alcohol (CH 3 OH).
-
In a chemical reaction, what is the theoretical yield and the percent yield?
-
In Exercises, find an equation of the tangent line to the graph of y = f (x) at the given x. Do not apply formula (6), but proceed as we did in Example 4. Example 4. Finding the Equation of the...
-
Paul sells two types of sweets at school. One cost R0,50 and the other cost R1,5. If she sells 300 sweets in total and makes R200 revenue. How much money was collected from the R0.50 sweets and how...
-
Did you know that, prior to the global AICPA initiatives, each country had its own version of accounting guidelines. Some were similar to US Generally Accepted Accounting Principles (GAAP) and some...
-
Explain at least 5 attributes/biases of labour market in Canada and how they can affect the duties of the HR manager and what are the implications.
-
The Carthage Corporation is currently (1/1/2018) considering the acquisition of the stock of LaGoulette Snowmobiles Inc. The financial statements for LaGoulette for the year ending 12/31/2017 are:...
-
In spur gears, the circle on which the involute is generated is called the (A) pitch circle (B) clearance angle (C) base circle (D) addendum circle
-
Based on the data presented in Exercise 4-23, illustrate the effects on the accounts and financial statements of Butler Co. for (a) The purchase, (b) The return of the merchandise for credit, and (c)...
-
Pearson Education, a publisher of college textbooks, would like to know if students prefer traditional textbooks or digital textbooks. A random sample of students was asked their preference and the...
-
If additional Cl 2 (g) is added to the reaction system at constant total pressure and temperature, how will the partial pressures of H 2 (g) and HCl(g) change?
-
Show that [(A/T)/(1/T)] V / U Write an expression analogous to Equation (6.36) that would allow you to relate A at two temperatures.
-
If additional Cl 2 (g) is added to the reaction system at constant V and T, how will the degree of dissociation of HCl(g) change
-
What are some practical advantages to using a star schema data model over a relational model in healthcare?
-
A contractor has to move 15 300 Bank m 3 of wet sandy material in rear dump trucks, which will be loaded by an excavator. Average face depth will be 2.4 m with 60-90 degree average swing angle. Ten...
-
1. Tony stark is trying to talk pepper Potts into attending an event with him. Pepper does not want to attend because she feels that she and will be much older than most of the younger crowd...

Study smarter with the SolutionInn App


