Use formal charge to identify the better Lewis structure. H H-C= H H=C
Question:
Use formal charge to identify the better Lewis structure.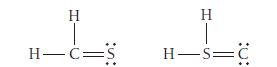
Transcribed Image Text:
H H-C= H H=C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
H HCS 00 ...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A long conducting wire made of anti-matter carries a uniform current per unit area. The anti-electrons (each carries charge +1.602 x 10-19 C ) with speed 4.862 m/s (the speed and magnitude of the...
-
Use formal charges to identify the better Lewis structure. H H-S-C-H 1 H H T H-C-S-H I H
-
LMO Company incurred expenditures for leasehold improvements during the year. The relevant lease is on an office building that the Company rented at the beginning of 2021. The improvements cost...
-
Consider the many moments of joy in the movie. Why include them? What risky acts of courage do Katherine, Mary and Dorothy take? How are they rewarded? The film shows the strong relationships that...
-
What is online analytical processing? How is OLAP related to databases? What is a pivot table, and how are pivot tables and OLAP related?
-
Equipment with a cost of $189,000 has an estimated residual value of $24,000, has an estimated useful life of 15 years, and is depreciated by the straight-line method. (a) Determine the amount of the...
-
A \(\$ 100,000\) bond priced at 103.5 can be bought or sold for a. \(\$ 100,000+\) interest b. \(\$ 103,500\) c. \(\$ 3,500\) d. \(\$ 10,350\)
-
Read this article on "neither leading nor ceding" and then respond to the following: do you agree or disagree with authors? Respond with your reasons for doing so. What did you learn from it?
-
5. Using the online exchange estimator, fill out the below table assuming that you insure just yourself. Use an income of $100. List the costs for the bronze, silver, and gold levels. Level Bronze...
-
In N 2 O, nitrogen is the central atom, and the oxygen atom is terminal. In OF 2 , however, oxygen is the central atom. Use formal charges to explain why. Formal charge = number of valence electrons...
-
Write a Lewis structure that obeys the octet rule for each ion. Include resonance structures if necessary and assign formal charges to each atom. a. CIO3 b. ClO4 c. NO3 + d. NH4
-
A heater coil connected to a 240-V rms ac line has a resistance of 34 . (a) What is the average power used? (b) What are the maximum and minimum values of the instantaneous power?
-
Consider that you have a recorder that is 44 cm long, and that when all finger holes are covered it plays an "F4 " that as you can see in the chart has a frequency of 349 Hz . ( the middle column is...
-
Your friend must make a $1,000 charge on his credit card to pay for car repairs and can only make the minimum payment for the next 3 months. Assume an APR of 15.99% and a required minimum payment of...
-
Suppose an investor faces a risk x that is uniformly distributed over [-1,1]. She is exposed to another risk y (on her labor income) that is independent from x . The payoffs of y are distributed as:...
-
Value = $370,000 Balance = $280,000 Current payment = $2,450 Interest Rate = 6.375% Closing Costs = 2.5% Prepaids = 1% What is the new loan amount/P&I payment/LTV?
-
You are evaluating two passive equity funds Vanguard Small-growth ETF (VBK) and Invesco Trust (QQQ). Both funds' objective is to track the performance of small and growth firms. The information about...
-
For 2009, PMD Inc. had set the following standards for production of metal tables: 35 pounds of iron at a standard cost of $2.60 per pound. During June, the company produced 300 tables. The company...
-
For liquid water the isothermal compressibility is given by; where r and b are functions of temperature only. If 1 kg of water is compressed isothermally and reversibly from I to 500 bar at 60(C. how...
-
What is the barometric pressure reading in millimeters of mercury corresponding to 101.325 kPa(abs)?
-
Why must a barometric pressure reading be corrected for temperature?
-
By how much would the barometric pressure reading decrease from its sea-level value at an elevation of 1250 ft?
-
You work as a bookkeeper for a small production company. Their accounting systems are not automated and, for this reason, they often make mistakes. You are to identify and correct the errors in the...
-
1) If shares are trading at $6.00 per share what is the Dividend Yield (%) on this stock? 2) How much Free Cash Flow did this company generate in 2022? 3) How many times did Accounts Receivable...
-
How health care is provided in ONE of these major industrialized nations: Germany, Canada, France, Brazil, Japan and describe their system. Who is covered, how is it paid for, what is and isn't...

Study smarter with the SolutionInn App


