Most of the second row transition metals do not follow the normal orbital filling pattern. Five of
Question:
Most of the second row transition metals do not follow the normal orbital filling pattern. Five of them—Nb, Mo, Ru, Rh, and Ag—have a [Kr] 5s14dx configuration and Pd has a [Kr] 4d10 configuration.
Write the ground state electron configuration for each species.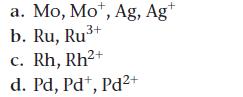
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





