Nomex, a condensation copolymer used by firefighters because of its flame-resistant properties, forms from isophthalic acid and
Question:
Nomex, a condensation copolymer used by firefighters because of its flame-resistant properties, forms from isophthalic acid and m-aminoaniline. Draw the structure of the dimer.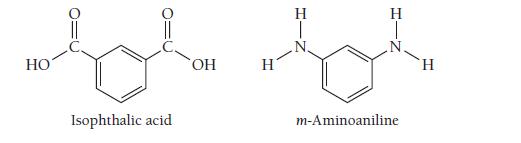
Transcribed Image Text:
HO Isophthalic acid OH H H T H m-Aminoaniline H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Chemical reaction between isophthal...View the full answer

Answered By

Vikash singh
i would like to solve the problems starting from basic to higher level.
Everything question having logic i would like to explore that logic and solve the question in short way in short time.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The polyester named Lactomer® is an alternating copolymer of lactic acid and glycolic acid. Lactomer is used for absorbable suture material because stitches of Lactomer hydrolyze slowly over a...
-
Draw the condensed structure of the compounds formed by condensation reactions between (a) benzoic acid and ethanol, (b) ethanoic acid and methylamine, (c) acetic acid and phenol. Name the compound...
-
The total pressure of vapor over liquid acetic acid at 71.3°C is 146 mmHg. If the density of the vapor is 0.702 g/L, what is the mole fraction of dimer in the vapor? See Problem 11.149. Problem...
-
Jansen, Inc., is a defense contractor that uses job costing. Because the firm uses a perpetual inventory system, the three supporting schedules to the income statement (the schedule of raw materials...
-
First Development Corporation of Kentucky (FDCK) sought to purchase a fifteen-acre parcel of riverfront property owned by Martin Marietta. On May 9, FDCK made an offer to purchase the property for...
-
Do the relationships between performance and satisfaction suggested by Porter and Lawler seem valid? Cite examples that both support and refute the model.
-
Founded in 1966 as a trader of oil and oil products, Vitol is a company with no external shareholders. All shareholders are also employees. It is a conglomerate company of energy companies that work...
-
Company is a small but growing manufacturer of telecommunications equipment. The company has no sales force of its own; rather, it relies completely on independent sales agents to market its...
-
Show that the mean square estimate is the mean of the posterior den- sity. Show this by differentiating eq. (13) at the lecture slides with respect to and set the obtained gradient to zero. Now the...
-
Polyacetylene is an addition polymer with the structure shown here. Draw the structure of the monomer. H [] C. C H
-
One kind of polyester is a condensation copolymer formed from terephthalic acid and ethylene glycol. Draw the structure of the dimer. HO Terephthalic acid OH HD-CH-CH-OH Ethylene glycol
-
(a) Let V = x 2 y 2 z in a region ( = 2 0 ) defined by 1 < x, y, z < 1. Find the charge density v in the region. (b) If the charge travels at 10 4 ya y m/s, determine the current crossing surface 0...
-
What is the best thing a programmer can do to improve memory access time? How does it improve memory access time?
-
Using an example identify three strategies that organisations may use to improve work-life balance among their employees? Which are likely to prove most effective and why? In this question you need...
-
Develop and justify a Professional Development Plan that will facilitate an increased level of digital skill attainment suitable for effective management and leadership in the contemporary workplace.
-
Marie Cole wants to be a manager. She enjoyed every course taught at school, and managed to score high grades. Each of these provided her with some clear-cut answers. Now the professor in her...
-
What is HIPPA? Why is it relevant to the practice of professional counseling? What does it require?
-
Velocity consulting firm enters into a contract to help Burger Boy, a fast-food restaurant, design a marketing strategy to compete with Burger King. The contract spans eight months. Burger Boy...
-
Draw the major product for each of the following reactions: (a) (b) (c) 1) 9-BBN 2) H2O2, NaOH 1) Disiamylborane 2) H20, NaOH
-
The three lowest energy levels for atomic carbon (C) have the following energies and degeneracies: What is the contribution to the average molar energy from the electronic degrees of freedom for C...
-
The lowest four energy levels for atomic vanadium (V) have the following energies and degeneracies: What is the contribution to the average energy from electronic degrees of freedom for V when T =...
-
Consider the following table of diatomic molecules and associated rotational constants: a. Calculate the rotational temperature for each molecule. b. Assuming that these species remain gaseous at 100...
-
Show the minimal network architecture in deep learning by organizing the nodes in each layer horizontally and specify weights and bias including calculations explanations for each node. x2 -1 -1 -1...
-
Write a program (using C program) to simulate a simple lottery engine. The user must enter 5 integer numbers, within the range of 10 to 30. The lottery engine must validate that all the numbers...
-
Entities and their attributes: Player Attributes: PlayerID (Primary Key), Username Character Attributes: CharacterID (Primary Key), PlayerID (Foreign Key), CharacterName, Team, SkillLevel Inventory...

Study smarter with the SolutionInn App


