Predict the products and write a balanced molecular equation for each reaction. If no reaction occurs, write
Question:
Predict the products and write a balanced molecular equation for each reaction. If no reaction occurs, write “NO REACTION.”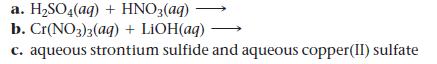
Transcribed Image Text:
a. H₂SO4(aq) + HNO3(aq) b. Cr(NO3)3(aq) + LiOH(aq) - c. aqueous strontium sulfide and aqueous copper(II) sulfate
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
ANSWER a H2SO4aq HNO3aq ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the products and write a balanced molecular equation for each reaction. If no reaction occurs, write NO REACTION. a. HCl(aq) + Hg2(NO3)2(aq) b. KHSO 3(aq) + HNO3(aq) - c. aqueous ammonium...
-
Magnesium carbonate, magnesium oxide, and magnesium hydroxide are all white solids that react with acidic solutions. (a) Write a balanced molecular equation and a net ionic equation for the reaction...
-
Assuming that each double-replacement reaction occurs, predict the products and write each balanced chemical equation. a. Zn(NO3)2 + NaOH ( ? b. HCl + Na2S ( ?
-
Reference groups influence us in three ways. These influences include informational, utilitarian, and dimensions. a. knowledge O b. O c. reputational O d. descriptive value-expressive
-
What is the difference between logical access to the computer and physical access to the computer? Why is the security of both important?
-
Lindon Company is the exclusive distributor for an automotive product that sells for $40 per unit and has a CM ratio of 30%. The companys fixed expenses are $180,000 per year. The company plans to...
-
Consider the net cash flows and salvage values shown below. Assume the alternatives can be indefinitely renewed with the same cash flows and salvage values. Using a MARR of 8%, specify the planning...
-
Imagine that Howard has asked you to write some queries to help him make better use of his data. For each information request below, write a single query that provides the answer set. When a task...
-
What happened to the overhead allocation numbers? Why aren't they included?
-
Hard water often contains dissolved Ca 2 + and Mg 2 + ions. One way to soften water is to add phosphates. The phosphate ion forms insoluble precipitates with calcium and magnesium ions, removing them...
-
Toilet bowl cleaners often contain hydrochloric acid, which dissolves the calcium carbonate deposits that accumulate within a toilet bowl. What mass of calcium carbonate (in grams) can 3.8 g of HCl...
-
What is the performance analysis of an algorithm?
-
Describe one specific control for each of the following categories of personnel control plans: hiring, retention, development, management, and termination.
-
What roles can an accountant play in the development or acquisition of an AIS?
-
Go to http://investor.kelloggs.com/annuals.cfm, to access the Kelloggs 2010 Annual Report, and find the statement of earnings. Sales are the revenue for a merchandise company. How much did Kelloggs...
-
What process is used to monitor controls and events to provide assurance in real-time? Why has the internal auditor been the main driver of the process?
-
What is the systems development life cycle (SDLC) methodology?
-
The December 31, 2009 balance sheet of the Blackmon Corporation disclosed the following information relating to its receivables: *The company is contingently liable for a discounted note receivable...
-
What is master production scheduling and how is it done?
-
Air at 40C. Appendix D gives dynamic viscosity for a variety of fluids as a function of temperature. Using this appendix, give the value of the viscosity for the following fluids
-
Hydrogen at 40C. Appendix D gives dynamic viscosity for a variety of fluids as a function of temperature. Using this appendix, give the value of the viscosity for the following fluids
-
Glycerin at 40C. Appendix D gives dynamic viscosity for a variety of fluids as a function of temperature. Using this appendix, give the value of the viscosity for the following fluids
-
Assume that the swap rate for an interest-rate swap is 7% and that the fixed-rate payments are made semiannually on an actual / 360 basis. If the notional amount of a two-year swap is $20 million in...
-
Suppose that an investor has a 6-year investment horizon. The investor is considering a 13-year 9% coupon bond selling at its par value of $1,000. The investor's expectations are as follows: 1. The...
-
You borrow $25,000 for a home repair. You can afford to pay back $800 a month. If you borrow the money directly from a bank, you have a 6.25% interest rate. How longwill it take you to pay off the...

Study smarter with the SolutionInn App


