Predict the products and write a balanced molecular equation for each reaction. If no reaction occurs, write
Question:
Predict the products and write a balanced molecular equation for each reaction. If no reaction occurs, write “NO REACTION.”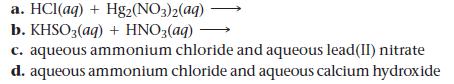
Transcribed Image Text:
a. HCl(aq) + Hg2(NO3)2(aq) b. KHSO 3(aq) + HNO3(aq) - c. aqueous ammonium chloride and aqueous lead(II) nitrate d. aqueous ammonium chloride and aqueous calcium hydroxide
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
a 2 HClaq Hg2NO32aq bKHSO3aq H...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the products and write a balanced molecular equation for each reaction. If no reaction occurs, write NO REACTION. a. HSO4(aq) + HNO3(aq) b. Cr(NO3)3(aq) + LiOH(aq) - c. aqueous strontium...
-
Magnesium carbonate, magnesium oxide, and magnesium hydroxide are all white solids that react with acidic solutions. (a) Write a balanced molecular equation and a net ionic equation for the reaction...
-
Assuming that each double-replacement reaction occurs, predict the products and write each balanced chemical equation. a. Zn(NO3)2 + NaOH ( ? b. HCl + Na2S ( ?
-
TRUE OR FALSE. 6 POINTS EACH. (Always assume "Other things equal.") 1. In economics in the short run, a firm will have both fixed and variable resources. 2. In economics, the short run is defined as...
-
Jean & Joan Cosmetics has a complete line of beauty products for women and maintains a computerized inventory system. An eight-digit product number identifies inventory items, of which the first four...
-
Outback Outfitters sells recreational equipment. One of the companys products, a small camp stove, sells for $50 per unit. Variable expenses are $32 per stove, and fixed expenses associated with the...
-
The cash flows associated with a project are shown below. The interest rate varies from year to year as shown. Determine an equivalent uniform annual series of cash flows. EOY Cash Flow Interest...
-
On the basis of your answers to Problems 25-1 and 25-2, if Hastings were to acquire Vandell, what would be the range of possible prices that it could bid for each share of Vandell common stock?...
-
Who is Tesla's auditor? Did Tesla's receive a "clean" (unmodified) audit opinion? How many critical audit matters were discussed in Tesla's audit report?
-
Toilet bowl cleaners often contain hydrochloric acid, which dissolves the calcium carbonate deposits that accumulate within a toilet bowl. What mass of calcium carbonate (in grams) can 3.8 g of HCl...
-
People sometimes use sodium bicarbonate as an antacid to neutralize excess hydrochloric acid in an upset stomach. What mass of hydrochloric acid (in grams) can 2.5 g of sodium bicarbonate neutralize?
-
Aut Q R is the identity group. Since every positive element of R is a square, it follows that an automorphism of R sends positives to positives and hence that it preserves the order in R. Trap a...
-
What does the approved configuration plan specify?
-
What are the four stages through which a program should move as it is being developed? Who should have responsibility for each of those phases?
-
What are the approaches to obtaining an RFP?
-
What is systems selection?
-
Research inventory fraud cases, and write a paper on a case you find interesting (your instructor will provide guidance regarding the papers length). At a minimum, you should include the following...
-
The 2011 audit of the accounting records of the Lane Company discloses the following information: Required 1. Reconstruct the journal entries that were made by Lane during 2011 to record changes in...
-
Review Exhibit 11.4. Analyze each product on the graph according to the characteristics that influence the rate of adoption. For example, what can you conclude from the data about the relative...
-
Air at 40F. Appendix D gives dynamic viscosity for a variety of fluids as a function of temperature. Using this appendix, give the value of the viscosity for the following fluids
-
Hydrogen at 40F. Appendix D gives dynamic viscosity for a variety of fluids as a function of temperature. Using this appendix, give the value of the viscosity for the following fluids
-
Glycerin at 60F. Appendix D gives dynamic viscosity for a variety of fluids as a function of temperature. Using this appendix, give the value of the viscosity for the following fluids
-
Cullumber Printing Corp. uses a job-order cost system. The following data summarize the operations related to the first quarter's production: 1. Materials purchased on account were $192,900, and...
-
The Fairfax location's regional manager wants to know the maximum of the units sold per order, and the minimum FOB $. Use functions to find these values. Date Agent Item Description Quantity FOBS...
-
Please fill in the yellow cells Fiscal Year --> 2021 WALMART, INC. - COMPANY VALUATION (Unaudited; USD 1000) 2022 2023 2024 2025 2026 TOTAL REVENUES Growth Rate vs Prior Year Total Operating Expenses...

Study smarter with the SolutionInn App


