Refer to the K sp values in Table 18.2 to calculate the molar solubility of each compound
Question:
Refer to the Ksp values in Table 18.2 to calculate the molar solubility of each compound in pure water.
a. MX (Ksp = 1.27 * 10-36)
b. Ag2CrO4
c. Ca(OH)2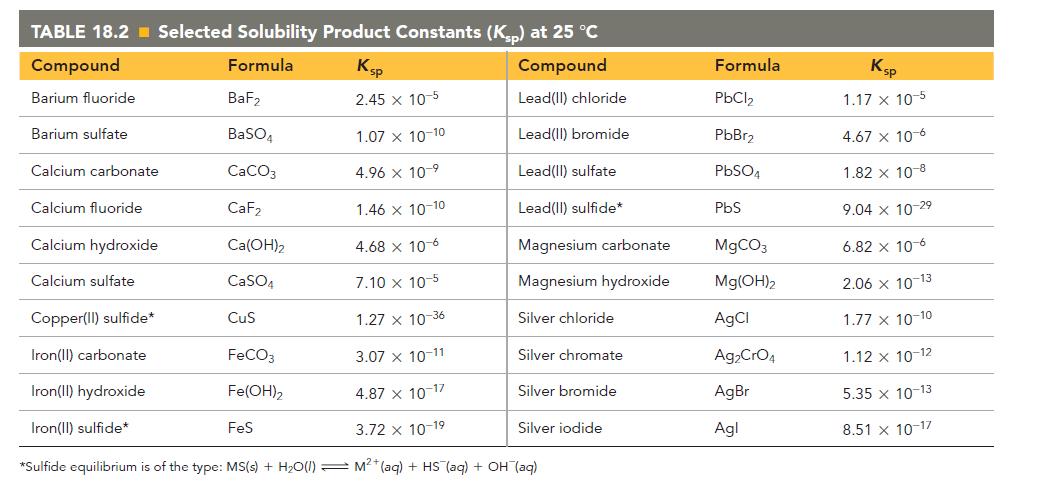
Transcribed Image Text:
TABLE 18.2 Selected Solubility Product Constants (Ksp) at 25 °C Compound Formula Ksp Compound Barium fluoride 2.45 x 10-5 Lead(II) chloride Lead(II) bromide Lead(II) sulfate Lead(II) sulfide* Barium sulfate Calcium carbonate Calcium fluoride Calcium hydroxide Calcium sulfate BaF₂ BaSO4 CaCO3 CaF₂ Ca(OH)₂ CaSO4 1.46 x 10-10 4.68 x 10-6 7.10 x 10-5 Copper(II) sulfide* 1.27 x 10-36 Iron(II) carbonate 3.07 x 10-11 Iron(II) hydroxide 4.87 x 10-17 Iron(II) sulfide* 3.72 x 10-19 *Sulfide equilibrium is of the type: MS(s) + H₂O(1) M²+ (aq) + HS (aq) + OH(aq) CuS FeCO3 Fe(OH)2 1.07 x 10-10 4.96 x 10-⁹ FeS Magnesium carbonate Magnesium hydroxide Silver chloride Silver chromate Silver bromide Silver iodide Formula PbCl₂ PbBr₂ PbSO4 PbS MgCO3 Mg(OH)2 AgCl Ag₂CrO4 AgBr Agl Ksp 1.17 x 10-5 4.67 x 10-6 1.82 x 10-8 9.04 x 10-2⁹ 6.82 x 10-6 2.06 x 10-13 1.77 x 10-10 1.12 x 10-12 5.35 x 10-13 8.51 x 10-17
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
To calculate the molar solubility of each compound in pure water we need to use the Ksp expression K...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Refer to the K sp values in Table 18.2 to calculate the molar solubility of each compound in pure water. a. AgBr b. Mg(OH) 2 c. CaF 2 TABLE 18.2 Selected Solubility Product Constants (Ksp) at 25 C...
-
Compound Ksp Iron(II) sulfide 3.7210-19 Magnesium hydroxide 2.0610-13 Lead(II) bromide 4.6710-6 A) Use the ksp values in the table to calculate the molar solubility of FeS in pure water. S= ____M...
-
Refer to Table 10.1 in the text and look at the period from 1973 through 1978. a. Calculate the arithmetic average returns for common stocks and T-bills over this period. b. Calculate the standard...
-
Consider the following table of countries and their associated maximum production possibilities for wheat and wool below. Note that these numbers represent tonnes of output per day. Country Wheat...
-
Alameda Service Center just purchased an automobile hoist for $15,000. The hoist has a 5-year life and an estimated salvage value of $1,080. Installation costs were $2,900, and freight charges were...
-
1. Why do you think Starbucks has been so concerned with social responsibility in its overall corporate strategy? 2. Is Starbucks unique in being able to provide a high level of benefits to its...
-
What is a compilation unit?
-
Samuel Aldrich started his own delivery service, Aldrich Service Inc., on June 1, 2012. The following transactions occurred during the month of June. June 1 Stockholders invested $15,000 cash in the...
-
Consider the Solow model we studied in class, with the production function given by: Y =AKN1. tttt In this question, we distinguish between the labor force (L) and the total population (Pop). The...
-
Write balanced equations and expressions for K sp for the dissolution of each ionic compound. a. CaCO 3 b. PbCl 2 c. AgI
-
Methyl red has a pK a of 5.0 and is red in its acid form and yellow in its basic form. If several drops of this indicator are placed in a 25.0-mL sample of 0.100 M HCl, what color will the solution...
-
The graph with Problem 24 shows vx(t) for a skateboard moving along the x-axis. Rank the times t = 0.5 s, 1.5 s, 2.5 s, 3.5 s, 4.5 s, and 5.5 s, in order of the magnitude of the acceleration, from...
-
Explain the concept of Total Quality Management (TQM) and its significance in contemporary business environments. Discuss the fundamental principles and components of TQM, emphasizing its focus on...
-
A fixed interest security pays coupons of 5 % per annum convertible half - yearly in arrears. The security is redeemable at 1 1 0 % at the option of the borrower on any anniversary date between 1 5...
-
A trunk that moves leftward by 3.13 m over a frictionless floor. 3 N and the indicated angle is 0 = 60. (a) During the displacement, v, the gravitational force, and the normal force? (b) Is there a...
-
A cupcake recipe designed to produce 2 6 cupcakes calls for 3 4 0 grams of flour. Determine the quantity of flour that would be required to make 3 0 cupcakes.
-
Select accounts of Burger Express are shown below as of December 31, 20X9, before any accounts have been adjusted for the current fiscal year. Prepaid rent 103,680 Accumulated depreciation - Van...
-
From the e-Activity, explain what either Lenski or Durkheim would say about the technologys impact of society. Draw upon your camping and/or hunting experience or television viewing of such shows as...
-
Modify the counter from Exercise 5.44 such that the counter will either increment by 4 or load a new 32-bit value, D, on each clock edge, depending on a control signal Load. When Load = 1, the...
-
What are the two chief ions found in seawater?
-
Seawater freezes at a lower temperature than pure water because of the salts dissolved in it. How does the boiling point of seawater compare with that of pure water?
-
What is the easiest way to distinguish between a solution that contains Cu 2+ ions from one that contains Ca 2+ ions?
-
3. Find adj (A), det (A), and A-1 if 20 3 A = 0 3 2 -2 0-4
-
After the 2019 Australia Bushfires, "A survey conducted in January 2020 found that 26% of Australian businesses had already been affected by the bushfires". Assume Mallacoota, a region of Australia...
-
Solve with the help of partial fraction Question 4: 2x-x+1 (x+3)(x-1)(x+5) Solve with the help of partial fraction Question 5: (x*)(x+1) Solve the help of partial fraction 4+7x (2+3x)(x+1)

Study smarter with the SolutionInn App


