Refer to the K sp values in Table 18.2 to calculate the molar solubility of each compound
Question:
Refer to the Ksp values in Table 18.2 to calculate the molar solubility of each compound in pure water.
a. AgBr
b. Mg(OH)2
c. CaF2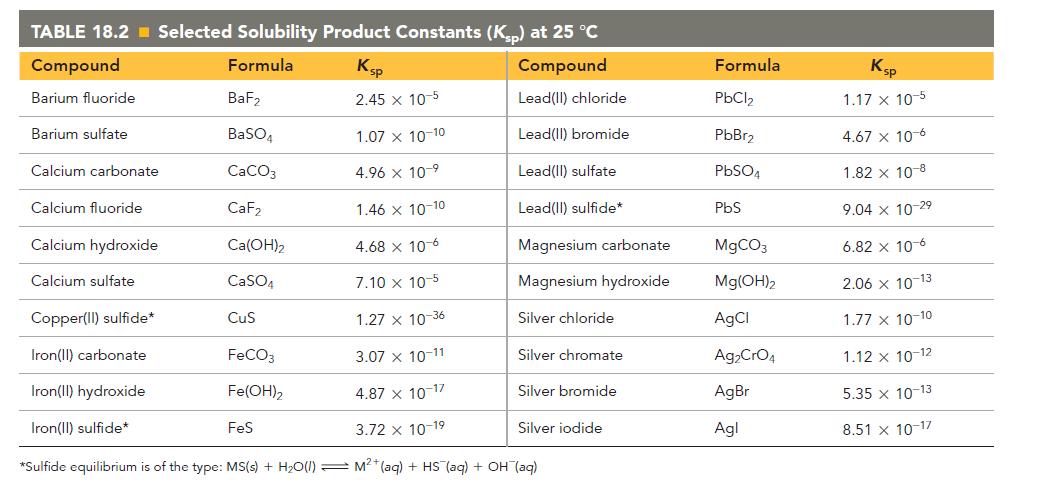
Transcribed Image Text:
TABLE 18.2 Selected Solubility Product Constants (Ksp) at 25 °C Compound Formula Ksp Compound Barium fluoride 2.45 x 10-5 Lead(II) chloride Lead(II) bromide Lead(II) sulfate Lead(II) sulfide* Barium sulfate Calcium carbonate Calcium fluoride Calcium hydroxide Calcium sulfate BaF₂ BaSO4 CaCO3 CaF₂ 1.46 x 10-10 4.68 x 10-6 7.10 x 10-5 Copper(II) sulfide* 1.27 x 10-36 Iron(II) carbonate 3.07 x 10-11 Iron(II) hydroxide 4.87 x 10-17 Iron(II) sulfide* 3.72 x 10-19 *Sulfide equilibrium is of the type: MS(s) + H₂O(1) M²+ (aq) + HS (aq) + OH(aq) Ca(OH)₂ CaSO4 CuS FeCO3 Fe(OH)2 1.07 x 10-10 4.96 x 10-⁹ FeS Magnesium carbonate Magnesium hydroxide Silver chloride Silver chromate Silver bromide Silver iodide Formula PbCl₂ PbBr₂ PbSO4 PbS MgCO3 Mg(OH)2 AgCl Ag₂CrO4 AgBr Agl Ksp 1.17 x 10-5 4.67 x 10-6 1.82 x 10-8 9.04 x 10-2⁹ 6.82 x 10-6 2.06 x 10-13 1.77 x 10-10 1.12 x 10-12 5.35 x 10-13 8.51 x 10-17
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
a 731 10 ...View the full answer

Answered By

Vincent Omondi
I am an extremely self-motivated person who firmly believes in his abilities. With high sensitivity to task and operating parameters, deadlines and keen on instructions, I deliver the best quality work for my clients. I handle tasks ranging from assignments to projects.
4.90+
109+ Reviews
314+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Refer to the K sp values in Table 18.2 to calculate the molar solubility of each compound in pure water. a. MX (Ksp = 1.27 * 10 -36 ) b. Ag 2 CrO 4 c. Ca(OH) 2 TABLE 18.2 Selected Solubility Product...
-
Compound Ksp Iron(II) sulfide 3.7210-19 Magnesium hydroxide 2.0610-13 Lead(II) bromide 4.6710-6 A) Use the ksp values in the table to calculate the molar solubility of FeS in pure water. S= ____M...
-
Tooth enamel is composed of hydroxyapatite, whose simplest formula is Ca 5 (PO 4 ) 3 OH, and whose corresponding Ksp = 6.8 10 -27 .As discussed in the Chemistry and Life box on page 730, fluoride in...
-
An LNC can detect alterations in the record by Select one: a. Examining notes written in the margin b. Looking through late entries for detailed explanations c. Finding a doctor's explanation that is...
-
Murphy Company manufactures and sells three products. Relevant per unit data concerning each product are given below. Instructions (a) Compute the contribution margin per unit of the limited resource...
-
Do you believe that Bernard Madoff worked alone, or do you think he had help in creating and sustaining his Ponzi scheme? Would this represent a conflict of interest? The fraud perpetrated by Bernard...
-
How do you prevent name clashes when using header files?
-
Sullivan sold t-shirts with the name Boston Marathon and the year of the race imprinted on them. The Boston Athletic Association (BAA) sponsors and administers the Boston Marathon and has used the...
-
Find the range of the function defined by the equation and the given domain. f(x) = 4x3; domain= {0, 1, 2, 3}
-
Write balanced equations and expressions for K sp for the dissolution of each ionic compound. a. CaCO 3 b. PbCl 2 c. AgI
-
Methyl red has a pK a of 5.0 and is red in its acid form and yellow in its basic form. If several drops of this indicator are placed in a 25.0-mL sample of 0.100 M HCl, what color will the solution...
-
The following events occurred during the year ended 30 June 2016 for Plumber Limited. 1. Opening balance of accrued salaries was $10 000. Wages expense for the year was $630 000 and cash paid for...
-
A cyclist cycles 2 9 . 6 km in a direction 3 0 degrees north of east and then a further 1 1 . 6 km in a direction 2 5 degrees westbod north. using components, determine the magnitude of the resultant...
-
In what ways does the criminal justice system exert a disproportionate influence or exhibit a pronounced tendency to target individuals of color to a greater extent than their white counterparts?
-
Determine the magnitude if a man walks 3 2 . 5 km 4 5 deg north of east, and then walks due east 1 6 . 5 km .
-
The critical angle for a special type of glass in air is 30.5 . The index of refraction for water is 1.33. What is the critical angle if the glass is immersed in water? Answer in units.
-
Could you elucidate the variances that distinguish the collectives to which you are affiliated from other cohorts within your societal milieu? Furthermore, how do sociologists explicate the genesis...
-
Define Strategic Planning. Provide a brief summary of why it is/or is not necessary for homeland security organizations to conduct strategic planning? Provide a brief summary of what strategic...
-
What are the six activities involved in the physical supply/distribution system?
-
You have a solution that contains Ca 2+ ions and another that contains Na + ions. How would adding a solution that contains CO 2- 3 ionss enable you to tell which is which?
-
What is the difference between a molecular ion and a polar molecule?
-
Ordinary tap water tastes different after it has been boiled. Can you think of the reason why?
-
On 1/2/22, Dayjon established a stock appreciation rights plan, entitling executives to receive cash for the difference between the market price of common stock and pre-established price of $20 on...
-
Solve matrix by finding the values of x and y Question 2: Find A 232 33 -3-3-2 -21 -1 4 3x-2y=6 2x+y=11
-
What would be your arguments for supporting tariffs? Under what circumstance(s) would you not support tariffs? Address the concepts below. Define specific tariff, ad valorem tariff, and compound...

Study smarter with the SolutionInn App


