Referring to Table 18.1, pick an indicator for use in the titration of each base with a
Question:
Referring to Table 18.1, pick an indicator for use in the titration of each base with a strong acid.
a. CH3NH2
b. NaOH
c. C6H5NH2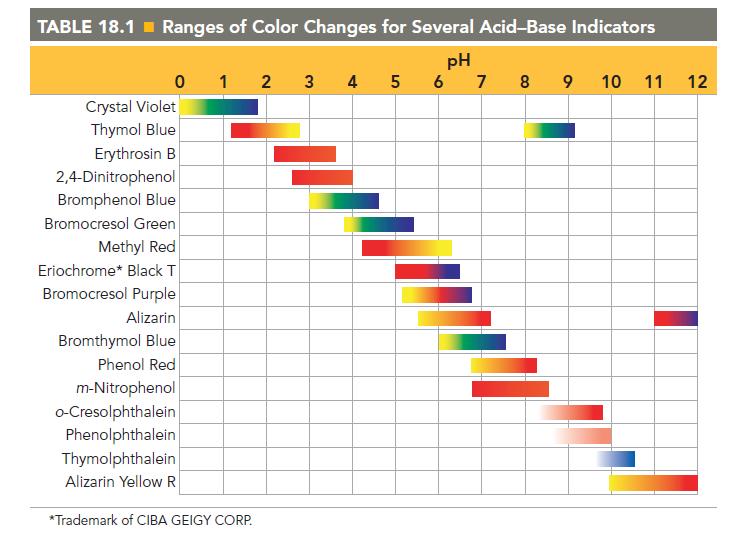
Transcribed Image Text:
TABLE 18.1 Ranges of Color Changes for Several Acid-Base Indicators pH 5 6 7 8 9 0 1 2 Crystal Violet Thymol Blue Erythrosin B 2,4-Dinitrophenol Bromphenol Blue Bromocresol Green Methyl Red Eriochrome* Black T Bromocresol Purple Alizarin Bromthymol Blue Phenol Red m-Nitrophenol o-Cresolphthalein Phenolphthalein Thymolphthalein Alizarin Yellow R *Trademark of CIBA GEIGY CORP. 3 4 tunnew 10 11 12
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
Based on Table 181the following indicators are appropriate for use in the titration of each base wit...View the full answer

Answered By

Cristine kanyaa
I possess exceptional research and essay writing skills. I have successfully completed over 5000 projects and the responses are positively overwhelming . I have experience in handling Coursework, Session Long Papers, Manuscripts, Term papers, & Presentations among others. I have access to both physical and online library. this makes me a suitable candidate to tutor clients as I have adequate materials to carry out intensive research.
4.90+
1538+ Reviews
3254+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Referring to Table 18.1, pick an indicator for use in the titration of each acid with a strong base. a. HF b. HCl c. HCN TABLE 18.1 Ranges of Color Changes for Several Acid-Base Indicators pH 5 6 7 8...
-
Weak base B has a pK b of 6.78 and weak acid HA has a pK a of 5.12. a. Which is the stronger base, B or A ? b. Which is the stronger acid, HA or BH + ? c. Consider the following reaction: B(aq) +...
-
Consider the curve shown here for the titration of a weak base with a strong acid and answer each question. a. What is the pH and what is the volume of added acid at the equivalence point? b. At what...
-
On February 1, 2020, Sheridan Company sells merchandise on account to Carla Vista Company for $6490. The entry to record this transaction by Sheridan Company is Sales Revenue Accounts Payable Notes...
-
Sara Casper, a recent graduate of Rollings accounting program, evaluated the operating performance of Klumpe Companys six divisions. Sara made the following presentation to the Klumpe board of...
-
How did the corporate culture of Enron contribute to its bankruptcy? Once upon a time, there was a gleaming headquarters office tower in Houston, with a giant tilted E in front, slowly revolving in...
-
List three ways to get input from the console and convert that input to the desired data type.
-
a. Prepare a plot of the CPI data. Based on this plot, which of the time series forecasting techniques covered in this chapter would not be appropriate for forecasting this time series? b. Apply...
-
Income elasticity Ei for food is about 0.30. Last year Ali spent $12,000 on food. If he gets a 4% raise in annual pay how much will he spent on food next year?
-
Methyl red has a pK a of 5.0 and is red in its acid form and yellow in its basic form. If several drops of this indicator are placed in a 25.0-mL sample of 0.100 M HCl, what color will the solution...
-
A 20.0-mL sample of a 0.125 M diprotic acid (H 2 A) solution is titrated with 0.1019 M KOH. The acid ionization constants for the acid are K a1 = 5.2 * 10 -5 and K a2 = 3.4 * 10 -10 . At what added...
-
Compare and contrast the two basic procurement strategies of corporate procurement and project procurement.
-
Explain the difference between procurement and strategic sourcing and outline the processes in making sourcing strategic?
-
Consider the Mass-spring system shown in Figure 1. (a) Find the equations of motion for the system, where my and m2 are the masses of the two objects, k, k2 and k3 are the spring constants of the...
-
What unexpected disparities might manifest in the realm of an individual's familial religious beliefs?
-
If the dart's exit speed is 1 4 . 0 m / sand the length of the blowgun is 1 . 4 0 m , find the time the dart is in the barrel.
-
Suppose corporate earnings are taxed at the corporate level (tax rate tc) and that earnings distributed to investors as income on equity are taxed privately (tax rate te) and earnings distributed to...
-
Commercial real estate prices and rental rates suffered substantial declines in the past year (Newsweek, July, 2009). These declines were particularly severe in Asia; annual lease rates in Tokyo,...
-
At the beginning of its fiscal year, Lakeside Inc. leased office space to LTT Corporation under a seven-year operating lease agreement. The contract calls for quarterly rent payments of $25,000 each....
-
From which class of solids would you expect electrons to be liberated most readily by the photoelectric effect when light is shined on a sample?
-
You are given two solids that look nearly alike, one of which is held together by ionic bonds and the other by van der Waals forces. How could you tell them apart?
-
What kind of solid is ice? Why does ice float when nearly all other solids sink when they freeze?
-
PHILOSOPHY OF ECONOMIC THOUGHT Individual Assignment Question Thomas Robert Malthus is an important figure in classical economic thought, but his ideas were rather controversial. Elaborate on the...
-
Introduction A. Introduce your childhood obesity and briefly explain what needs to change to address the issue. II. Barriers A. Identify two potential socioeconomic barriers to change and describe...
-
How Policy is Made, Energy Politics, and Nevada. Pause after the "Energy Politics" section. I need to find a news article on an energy topic of interest to you. (Put a link to the article below)....

Study smarter with the SolutionInn App


