Referring to Table 18.1, pick an indicator for use in the titration of each acid with a
Question:
Referring to Table 18.1, pick an indicator for use in the titration of each acid with a strong base.
a. HF
b. HCl
c. HCN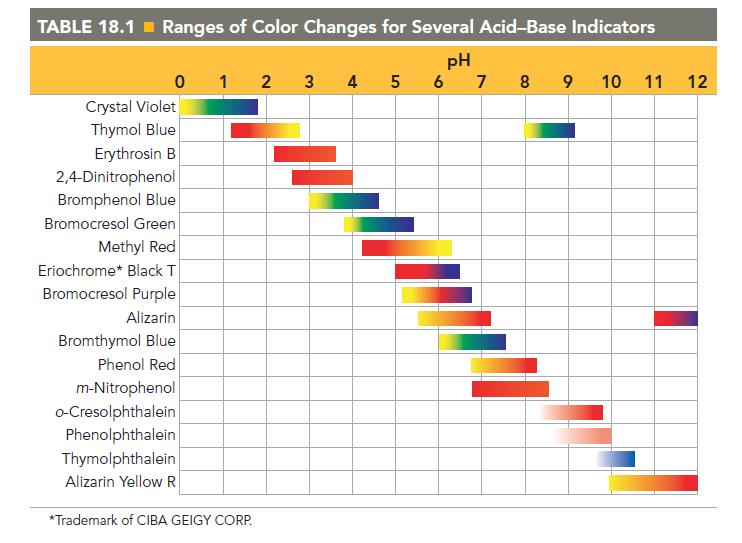
Transcribed Image Text:
TABLE 18.1 Ranges of Color Changes for Several Acid-Base Indicators pH 5 6 7 8 9 0 1 2 Crystal Violet Thymol Blue Erythrosin B 2,4-Dinitrophenol Bromphenol Blue Bromocresol Green Methyl Red Eriochrome* Black T Bromocresol Purple Alizarin Bromthymol Blue Phenol Red m-Nitrophenol o-Cresolphthalein Phenolphthalein Thymolphthalein Alizarin Yellow R *Trademark of CIBA GEIGY CORP. 3 4 tunnew 10 11 12
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a Phenol red mni...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Referring to Table 18.1, pick an indicator for use in the titration of each base with a strong acid. a. CH 3 NH 2 b. NaOH c. C 6 H 5 NH 2 TABLE 18.1 Ranges of Color Changes for Several Acid-Base...
-
Weak base B has a pK b of 6.78 and weak acid HA has a pK a of 5.12. a. Which is the stronger base, B or A ? b. Which is the stronger acid, HA or BH + ? c. Consider the following reaction: B(aq) +...
-
How does titration of a strong,monoprotic acid with a strong base differ from titration of a weak, monoprotic acid with a strong base with respect to the following: (a) Quantity of base required to...
-
Informative Speech Critique Evaluating Rhetorical Choices You may just number the questions and add a paragraph or two to answer - 1. Did you find the speaker credible? Why or why not? 2. Did the...
-
Bryant Bikes could sell its bicycles to retailers either assembled or unassembled. The cost of an unassembled bike is as follows. Direct materials .............. $150 Direct labor ............... 70...
-
Consider the two (excess return) index-model regression results for stocks A and B. The risk-free rate over the period was 6%, and the markets average return was 14%. Performance is measured using an...
-
What rules apply to service and filing of pleadings?
-
Selected transactions from the journal of Palm Tree Travel, Inc., are presented below. (a) Post the transactions to T accounts. (b) Prepare a trial balance at August 31,2008. Datc Account Titles and...
-
If a person gets legally drunk - meaning their BAC is 0.08% or higher; what will determine how long it takes for them to become sober? What is the role of physical and environmental factors...
-
A 20.0-mL sample of a 0.125 M diprotic acid (H 2 A) solution is titrated with 0.1019 M KOH. The acid ionization constants for the acid are K a1 = 5.2 * 10 -5 and K a2 = 3.4 * 10 -10 . At what added...
-
A 20.0-mL sample of 0.115 M sulfurous acid (H 2 SO 3 ) solution is titrated with 0.1014 M KOH. At what added volume of base solution does each equivalence point occur?
-
Suppose that \(X_{1}, \ldots, X_{n}\) are a set of independent and identically distributed random variables from a distribution \(F\) that has mean equal to zero, unit variance, and cumulant...
-
Via Gelato, a popular neighborhood gelato shop, provided the following cost formulas and actual results for the month of June: Fixed Element per Month Variable Element per Liter Actual Total for June...
-
You prepared for this assignment by completing this weeks discussion activity, in which you conducted a search of the research literature and wrote a draft literature review of four peer-reviewed...
-
line segments AB and CD are parallel. If the points are A ( 5 , - 4 ) , B ( - 3 , 2 ) , C ( 0 , 5 ) and D ( 4 , a ) , determine exact value for a
-
Grand River University Water Buffalo's case Explain Recommendations with Two Points 1. Who are the stakeholders? 2. What is the ABC breakdown? How do they assign the costs ? Should they keep all...
-
1. Analyze the following expression. Write down the factors, terms, the coefficients, constants and the operations. -2(4x4-2x+16) 2. Use what you have learned about exponents to write three different...
-
What is the purpose of personal financial statements, and how do they differ from those presented by an audited organization?
-
A city maintains a solid waste landfill that was 12 percent filled at the end of Year 1 and 26 percent filled at the end of Year 2. During those periods, the government estimated that total closure...
-
To fill in some of the details in the derivation of the smallest phase increment separating two resolvable FabryPerot fringes, that is, satisfy yourself that Show that Eq. (9.72) can be rewritten as...
-
Consider the interference pattern of the Michelson Interferometer as arising from two beams of equal flux density. Using Eq. (9.17), compute the half-width. What is the separation, in δ,...
-
Satisfy yourself of the fact that a film of thickness /4 and index n 1 will always reduce the reflectance of the substrate on which it is deposited, as long as n s > n 1 > n 0 . Consider the...
-
A boy pushes a 125 kg crate up a ramp inclined at 25o from the horizontal the floor by applying a force of 770 N parallel to the ramp. Calculate the coefficient of kinetic friction if the crate...
-
A copper wire that has been heated to 520C for pliability is plunged into 350 ml of water at 18C. The final temperature of the copper and water bath is 31 C. Find the mass of the copper wire....
-
A raccoon whacks a 30 g toy on a straight, horizontal floor that imparts to it an initial speed of v 0 = 5 m/s. The coefficient of kinetic friction between toy and floor is k = 0.14. What is the...

Study smarter with the SolutionInn App


