Sketch a voltaic cell for each redox reaction. Label the anode and cathode and indicate the half-reaction
Question:
Sketch a voltaic cell for each redox reaction. Label the anode and cathode and indicate the half-reaction that occurs at each electrode and the species present in each solution. Also indicate the direction of electron flow.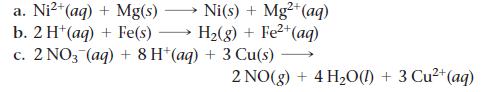
Transcribed Image Text:
2+ Ni(s) + Mg²+ (aq) H₂(g) + Fe²+ (aq) a. Ni²+ (aq) + Mg(s) b. 2 H+ (aq) + Fe(s) c. 2 NO3(aq) + 8 H+ (aq) + 3 Cu(s) 2 NO(g) + 4H₂O(l) + 3 Cu²+ (aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a Redox Reaction Niaq Mgs Nis Mgaq Voltaic Cell Anode Oxid...View the full answer

Answered By

Jehal Shah
I believe everyone should try to be strong at logic and have good reading habit. Because If you possess these two skills, no matter what difficult situation is, you will definitely find a perfect solution out of it. While logical ability gives you to understand complex problems and concepts quite easily, reading habit gives you an open mind and holistic approach to see much bigger picture.
So guys, I always try to explain any concept keeping these two points in my mind. So that you will never forget any more importantly get bored.
Last but not the least, I am finance enthusiast. Big fan of Warren buffet for long term focus investing approach. On the same side derivatives is the segment I possess expertise.
If you have any finacne related doubt, do reach me out.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Sketch a voltaic cell for each redox reaction. Label the anode and cathode and indicate the half-reaction that occurs at each electrode and the species present in each solution. Also indicate the...
-
Use line notation to represent each electrochemical cell in Problem 44. Problem 44 Sketch a voltaic cell for each redox reaction. Label the anode and cathode and indicate the half-reaction that...
-
Use line notation to represent each electrochemical cell in Problem 43. Problem 43 Sketch a voltaic cell for each redox reaction. Label the anode and cathode and indicate the half-reaction that...
-
Landry State University, a public university located in Louisiana, has a June 30 fiscal year. On July 20 of the current year, Landry State University receives $2,125,000 in payments from the U.S....
-
Prepare a flexible budget for 10,000, 12,000, and 14,000 units of output, using the following information: Variable costs Direct materials.......$10.00 per unit Direct labor...........$3.00 per unit...
-
What are the major accounting classifications in the world? What are the distinguishing features of each model?
-
The following data are for Marvin Department Store. The account balances (in thousands) are for 2017. 1. Compute (a) the cost of goods purchased and (b) the cost of goods sold. 2. Prepare the income...
-
Dousmann Corp.'s sales slumped badly in 2014. For the first time in its history, it operated at a loss. The company's income statement showed the following results from selling 500,000 units of...
-
How does a Cloud Data Management Platform differ from a Database Management System?
-
Calculate the standard cell potential for each of the electrochemical cells in Problem 43. Problem 43 Sketch a voltaic cell for each redox reaction. Label the anode and cathode and indicate the...
-
Balance each redox reaction occurring in basic aqueous solution. a. MnO4 (aq) + Br (aq) MnO(s) + BrO3(aq) b. Ag(s) + CN- (aq) + O(g) Ag(CN) (aq) c. NO (aq) + Al(s) - NH3(g) + AlO (aq)
-
If a ____ assumption is violated, a researcher can still proceed with the test as long as the violation is not too great.
-
Z Lemon Inc. is a women's shoe retailer. On February 1, 2022, the company purchased 500 pairs of "Angel" model shoes for $20 per pair. On March 3, the company purchased 700 pairs of the newly...
-
Delmond and Sons, LLC, owns an office which is insured under a Building and Personal Property Coverage Form (BPP).The new CEO of the company reduces the limit of insurance to $100,000 on the building...
-
DEF Company estimated the following cost for its only product, F-100: Per unit Selling price $19 Variable cost 6 Fixed cost 3 Fixed cost per unit is based on estimated production of 10,000 units....
-
In a few sentences, explain the targeted improvements to the accounting for derivatives and hedged issued by the FASB in 2017 which are currently effective for financial reporting purposes?
-
Does a tax preparer need to abide by any ethical guidance other than circular 230?
-
The capsize screening value C should be less than 2 if a boat is to be considered safe for ocean sailing. The formula is given as C = 4d-1/3b where d is the displacement in pounds and b is the beam...
-
Sue Deliveau opened a software consulting firm that immediately paid $2,000 for a computer. Was this event a transaction for the business?
-
A cargo ship travels from the Mississippi River into the Gulf of Mexico. Will the ship sink or rise with respect to the waterline as it moves from the river to ocean water? Why?
-
Consider an airplane window (area 0.30 m 2 ). If the pressure inside the plane is atmospheric pressure and the pressure outside is 20% of P atm , what is the force on the window? Ignore the velocity...
-
Three containers are filled with water to the same height (Fig. Q10.9). For which container is the pressure at the bottom the greatest? Or, are the pressures the same? Explain. Figure Q10.9 Container...
-
Ed Fathly placed $10,000 in a savings account every year for 6 years. The account paid 6% interest compounded annually. How much money will be in the account at the end of 6 years?
-
Tom and Henry were close friends, so after college they purchased a home together as tenants in common. They bought the home for $20,000 and 30 years later sold it for $600,000. How much of the...
-
Bonnie and Clyde get married and buy a home for $25,000. After 5 years, they file for divorce. As part of the divorce decree, Bonnie can live in the home until she reaches the age of 65, at which...

Study smarter with the SolutionInn App


