Sulfuric acid (H 2 SO 4 ) is a component of acid rain that forms when SO
Question:
Sulfuric acid (H2SO4) is a component of acid rain that forms when SO2, a pollutant, reacts with oxygen and water according to the simplified reaction:![]()
The generation of the electricity used by a medium-sized home produces about 25 kg of SO2 per year. Assuming that there is more than enough O2 and H2O, what mass of H2SO4, in kg, can form from this much SO2?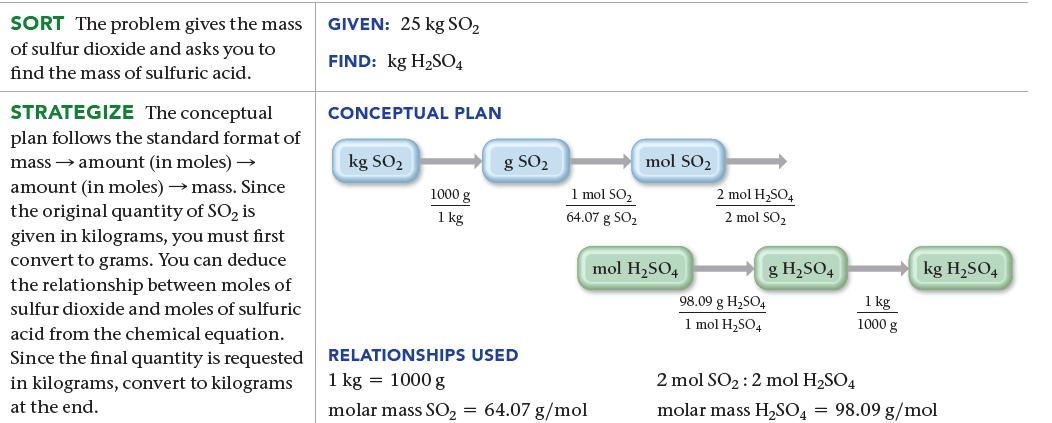
Transcribed Image Text:
2 SO₂(g) + O₂(g) + 2 H₂O(1) 2 H₂SO4(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
25 kg S X 1000 g 1 kg X 1 molSO 6407 g SO X 2 mol HSO4 2 molSO X 9809 ...View the full answer

Answered By

Vincent Omondi
I am an extremely self-motivated person who firmly believes in his abilities. With high sensitivity to task and operating parameters, deadlines and keen on instructions, I deliver the best quality work for my clients. I handle tasks ranging from assignments to projects.
4.90+
109+ Reviews
314+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Sulfuric acid (H2SO4) is a component of acid rain that forms when SO2, a pollutant, reacts with oxygen and water according to the simplified reaction: 2 SO2(g) + O2(g) + 2 H2O(l) -------> 2 H2SO4(aq)...
-
Nitric acid is a component of acid rain that forms when gaseous nitrogen dioxide pollutant reacts with gaseous oxygen and liquid water to form aqueous nitric acid. Write the balanced chemical...
-
Sulfuric acid is a component of acid rain formed when gaseous sulfur dioxide pollutant reacts with gaseous oxygen and liquid water to form aqueous sulfuric acid. Write the balanced chemical equation...
-
Researchers investigated the relationship between the number of involuntary admissions (detentions) for mental disorders a year under the Mental Health Act 1983 and the number of NHS psychiatric beds...
-
Data for Rivera Company are presented in P12-7B. Further analysis reveals the following. 1. Accounts payable pertains to merchandise creditors. 2. All operating expenses except for depreciation are...
-
A vertical, double-pane window, which is I m on a side and has a 25-mm gap filled with atmospheric air, separates quiescent room air at T ,o = 20C from quiescent ambient air at T ,o = - 20C....
-
Many researchers are interested in the transcription of protein-encoding genes in eukaryotes. Such researchers want to study mRNA. One method that is used to isolate mRNA is column chromatography....
-
Marston Corporation manufactures disposable thermometers that are sold to hospitals through a network of independent sales agents located in the United States and Canada. These sales agents sell a...
-
ESG CSR Study caseSunset Manufacturing Inc. manufactures Umbrellas in the Western Canadian market, and is comparing the budget for the current year with the actual results. The market has been good...
-
Jeff Smith realizes that the first thing he must do is compare the liquidity, leverage, activity, and profitability ratios of the two companies. Using the income statement and balance sheet data...
-
Identify the reactants and products in this chemical equation. 4 NH 3 (g) + 5 O2( g) 4 NO(g) + 6 H 2 O(g)
-
Manganese(IV) oxide reacts with aluminum to form elemental manganese and aluminum oxide: What mass of Al is required to completely react with 25.0 g MnO 2 ? a) 7.76 g Al b) 5.82 g Al c) 33.3 g Al d)...
-
What happens when a server uses a port to bind an object, but the port is not available? (a) The object is bound successfully. (b) The server is blocked until the port is available. (c) The server...
-
Introduction Talk about the history of Publix where everything started and how the company is still active now. 1-Describe Publix management system. 2-How Publix manage their inventory and warehouse?...
-
Explain to me how to get the manufacturing overhead number? Trees Sod Selling Price Per Unit $ 12.99 $ 0.38 Direct Materials Per Unit $ 0.99 $ 0.01 Direct Labor Per Unit $ 2.00 $ 0.09 Direct Labor...
-
Using the SEC 10-K report for Vertex pharmaceuticals please respond to the following: What is the name of the auditor? Are they one of the "Big Four"? conduct a search to see who the "Big Four" audit...
-
On 1 June 2023, Zul Ghani established a management consultancy firm, ZG Consultancy Sdn Bhd. During the month, Zul carried out the following transactions: June 1 Zul deposited RM25,000 into bank...
-
Your client is an individual who is the sole shareholder of a private corporation that is not a CCPC. The client expects the corporation's income and expenses for the coming year to be as follows:...
-
On July 31, 2012, the balances of the accounts appearing in the ledger of Vagabond Furnishings Company, a furniture wholesaler, are as follows: a. Prepare a multiple-step income statement for the...
-
On April 29, 2015, Auk Corporation acquires 100% of the outstanding stock of Amazon Corporation (E & P of $750,000) for $1.2 million. Amazon has assets with a fair market value of $1.4 million (basis...
-
Calculate the difference in pressure across the liquidair interface for a (a) Mercury and (b) Methanol droplet of radius 125 nm.
-
Calculate the vapor pressure of CH 3 OH(l) at 298.15 K if He is added to the gas phase at a partial pressure of 200. Bar using the data tables. By what factor does the vapor pressure change?
-
Use the vapor pressures of SO 2 (l) given in the following table to calculate the enthalpy of vaporization using a graphical method or a least squares fitting routine. T (K) 190. P (Pa) T (K) 230. P...
-
. Conduct research and review the pricing models for Amazon Web Services and Microsoft's Azure. . How are they similar and how are they different?
-
Two blocks are fastened to the ceiling of an elevator as in Figure. The elevator accelerates upward at 2.00 m/s. Find the tension in each rope. T 10.0 kg T 10.0 kg
-
Suzanne Michaels from Howie's Pizza was asked to calculate the break-even point for a new line of gourmet pizzas (prebaked, ready to take home). The selling price will be $25 per pizza. The labor...

Study smarter with the SolutionInn App


