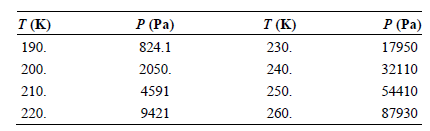
Use the vapor pressures of SO 2 (l) given in the following table to calculate the enthalpy
Question:
Transcribed Image Text:
T (K) 190. P (Pa) T (K) 230. P (Pa) 17950 32110 54410 87930 824.1 2050. 200. 240. 210. 250. 4591 220. 9421 260.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
A least squares fit of ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use the vapor pressures of ice given here to calculate the enthalpy of sublimation using a graphical method or a least squares fitting routine. T (K) P (Torr) 200. 0.1676 210. 0.7233 2.732 220. 230....
-
Use the vapor pressures of n-butane given in the following table to calculate the enthalpy of vaporization using a graphical method or a least squares fitting routine. P (Pa) 1000 x 104 1000 x 105 T...
-
Use the vapor pressures of tetrachloromethane given in the following table to calculate the enthalpy of vaporization using a graphical method or a least squares fitting routine. P/Pa T (K) 320. 330....
-
Design for Quality (D4Q) is a consulting firm that specializes in the design and implementation of quality management programs for service companies and organizations. It has had success designing...
-
Rewrite the following footnote based upon the GAAP of the country that you selected for your course project. Below is a footnote disclosure from AT&T's 2007 Annual Report regarding foreign currency...
-
On your own or with a classmate, choose one of the following products and create an advertisement that illustrates how your company creates time, place, and form utility in its delivery of the...
-
Alicin Nagle, an in-charge auditor, is reviving the inventory controls for the city of Carlton's vehicle maintenance facility. Nagle has learned the following: a. Vehicle maintenance records indicate...
-
Recall the Toms, Inc., problem. Letting W = jars of Western Foods Salsa M = jars of Mexico City Salsa Leads to the formulation: Max1W + 1.25M s.t. 5W + 7M 4480 Whole tomatoes 3W + 1M 2080 Tomato...
-
The City of Troy collects its annual property taxes late in its fiscal year. Consequently, each year it must finance part of its operating budget using tax anticipation notes. The notes are repaid...
-
Describe the role of technology in both Amazon's manufacturing and service operations.
-
When managers utilize a food cost percentage approach to menu item pricing they are basing their prices on the idea that food cost should be a. A predetermined percentage of selling price. b. Less...
-
Prove that a substance for which the solidliquid coexistence curve has a negative slope contracts upon melting.
-
Helicobacter pylori, the bacterium responsible for gastric ulcers, can survive in the stomach (where the pH is as low as 1.5) in part because it synthesizes large amounts of the enzyme urease. (a)...
-
Identify two digital marketing methods that can be used for brand awareness and support with reasons. Define short-term and long-term goals for brand awareness in the digital marketing strategy. How...
-
How much pressure is applied to the ground by a 56 kg man who is standing on square stilts that measure 0.06 m on each edge? Answer in units of Pa. 003 (part 2 of 2) 10.0 points What is this pressure...
-
Explain how marketing to different countries or cultures may affect your advertising message or campaign? Explain the principle and give examples of actual advertising campaigns localized for...
-
7. A rocket of a mass 200 kg loaded with a propellant of a mass 1200 kg is powered by a rocket engine whose exhaust velocity is 5000 kmph. What is the maximal fuel consumption per second possible,...
-
8. An airplane is flying N75E with an airspeed of 550 mph and a 45 mph wind blowing S75E. What is the actual speed, in mph and direction, written as a bearing, of the plane? Round to the nearest 1...
-
Evaluate the integral. dx
-
In Problems 1522, find the principal needed now to get each amount; that is, find the present value. To get $750 after 2 years at 2.5% compounded quarterly.
-
An object of mass m is subjected to a gradually increasing force given by F 0 (1e bt ) where a and b are constants. Solve the equation of motion of the particle. Find the particular solution for the...
-
The differential equation for a secondorder chemical reaction without back reaction is where c is the concentration of the single reactant and k is the rate constant. Set up an Excel spreadsheet to...
-
Obtain the solution for Eq. (12.62) for the forced harmonic oscillator using Laplace transforms.
-
This is for my Organizational Behavior class. This is due by midnight tonight, July 30. Please help!! The organization I have chosen is a rural health care facility...a clinic. What are the basic...
-
What are the functions of organizational culture? Use your own experience in an organization (workplace, school, gym, etc.) to explain how organizational culture contributes either positively or...
-
In 2 0 2 4 , Bratten Fitness Company made the following cash purchases: The exclusive right to manufacture and sell the X - Core workout equipment from Symmetry Corporation for $ 2 1 8 , 0 0 0 ....

Study smarter with the SolutionInn App


