The decomposition of ozone shown here is important to many atmospheric reactions: A study of the kinetics
Question:
The decomposition of ozone shown here is important to many atmospheric reactions:![]()
A study of the kinetics of the reaction results in the following data: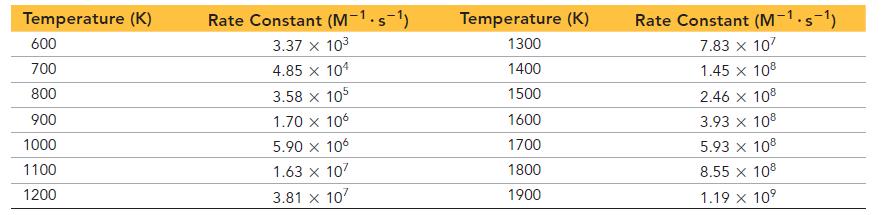
Determine the value of the frequency factor and activation energy for the reaction.
Transcribed Image Text:
03(g) O₂(g) + 0(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
To determine the frequency factor and activation energy prepare a graph of the natural ...View the full answer

Answered By

Arun kumar
made more than four thousand assignments
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The reaction between ethyl iodide and hydroxide ion in ethanol (C2H5OH) solution, C2H5I(alc) + OH (alc) -- C2H5OH(I) + I(alc), has an activation energy of and a frequency factor of 2.10 1011 M-1...
-
The decomposition of ozone is believed to occur in two steps: Identify any reaction intermediate. What is the overall reaction? 0302 O (elementary reaction) 20 (elementary reaction)
-
The following mechanism has been proposed to account for the rate law of the decomposition of ozone to O2(g): Apply the steady-state hypothesis to the concentration of atomic oxygen, and derive the...
-
An average of three small businesses go bankrupt each month. What is the probability that five small businesses will go bankrupt in a certain month?
-
Bill Johnson, sales manager, and Diane Buswell, controller, at Current Designs are beginning to analyze the cost considerations for one of the composite models of the kayak division. They have...
-
A company issues a $6,000,000, 12%, five-year bond that pays semiannual interest of $360,000 ($6,000,000 12% ), receiving cash of $6,463,304. Journalize the bond issuance.
-
Refer to the information from QS 21-18. Compute the variable overhead spending variance and the variable overhead efficiency variance and classify each as favorable or unfavorable. Data From QS 21-18...
-
The intangible assets section of Glover Company at December 31, 2013, is presented below. Patents ($60,000 cost less $6,000 amortization) ....... $54,000 Franchises ($48,000 cost less $19,200...
-
Explain the difference between " management controls ", which are generally the responsibility of the client's Management team and " transaction controls ", which are typically performed by...
-
How is the order of a reaction generally determined?
-
The reaction X products is second order in X and has a rate constant of 0.035 M -1 s -1 . If a reaction mixture is initially 0.45 M in X, what is the concentration of X after 155 seconds? a) 7.6 M...
-
Identify the regression objective of the following research questions (from the four options of estimating causal effects, forecasting/predicting an outcome, determining predictors of an outcome, and...
-
Job stress is a major problem for employees working in many organizations today. Discuss some of the job stressors. What can a manager do to reduce stressors for employees?
-
Mintzberg suggests that managerial roles should encompass interpersonal, decisional, and informational roles. Clearly, this is an idealized vision of the manager. They are encouraged to encompass all...
-
Technological developments deskill the global workforce. For example, factory-built, flat-pack furniture cut out the role of experienced carpenters. Similarly, with some vehicles having on-board...
-
In 2013, a clothing factory in Bangladesh collapsed, killing 1,138 people. Some 27 global brands, including Walmart and Benetton, were using the factory. One year on, these two corporations were...
-
Scott Emmons was working for Neiman Marcus, the luxury retailer, as an enterprise architect when he realized a big gap in how their stores were handling technology. Customers were showing up with...
-
Dona Corp. was started in 2013. The following summarizes transactions that occurred during 2013: 1. Issued a $40,000 face value discount note to Golden Savings Bank on April 1, 2013. The note had a 6...
-
A 2500-lbm car moving at 15 mi/h is accelerated at a constant rate of 15 ft/s 2 up to a speed of 50 mi/h. Calculate force and total time required?
-
A ship tows an instrument package in the form of a hemisphere with an open back at a velocity of 25.0 ft/s through seawater at 77F. The diameter of the hemisphere is 7.25 ft. Compute the force in the...
-
An antenna in the shape of a cylindrical rod projects from the top of a locomotive. If the antenna is 42 in long and 0.200 in. in diameter, compute the drag force on it when the locomotive is...
-
A bulk liquid transport truck incorporates a cylindrical tank 2 m in diameter and 8 m long. For the tank alone, calculate the pressure drag when the truck is traveling at 100 km/h in still air at 0C.
-
Please show all workings. Thank you. 3. (a) Assume the following information regarding locational arbitrage: Beta Bank Bid price of New Zealand dollar $0.406 Ask price of New Zealand dollar $0.409...
-
The mission, vision, and goals statements should Blank______. Multiple choice question. be shared with all stakeholders only be released in secured SEC documents be considered top-secret company...
-
Problem 13.63 The moon's radius is 1738 km. The magnitude of the acceleration due to gravity of the moon at a distances from the center of the moon is 4.89 1012 $2 m/s. Suppose that a spacecraft is...

Study smarter with the SolutionInn App


