The mass of fuel in a jet must be calculated before each flight to ensure that the
Question:
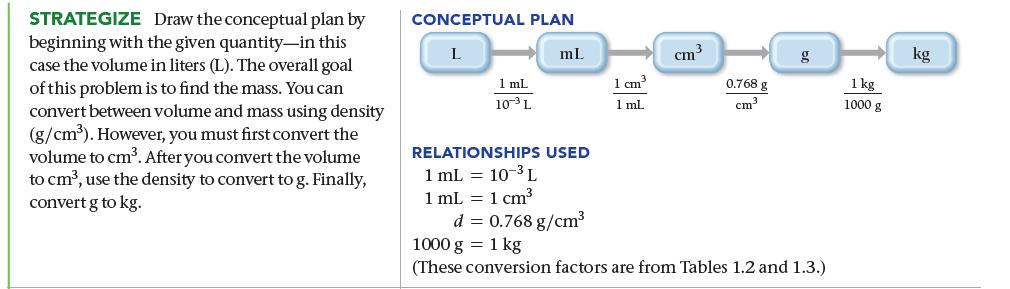
The mass of fuel in a jet must be calculated before each flight to ensure that the jet is not too heavy to fly. A 747 is fueled with 173,231 L of jet fuel. If the density of the fuel is 0.768 g/cm3, what is the mass of the fuel in kilograms?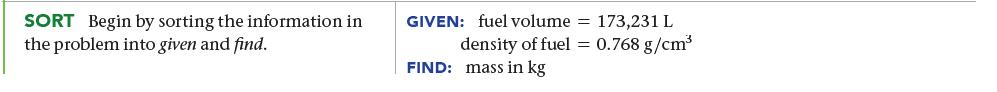
Transcribed Image Text:
SORT Begin by sorting the information in the problem into given and find. GIVEN: fuel volume = 173,231 L density of fuel = 0.768 g/cm³ FIND: mass in kg
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
173231 L X 1 ...View the full answer

Answered By

Benish Ahmad
I'm a professional software engineer. I'm lectutrer at GCUF and I have 3 years of teaching experience. I'm looking forward to getting mostly computer science work including:
Programming fundamentals
Object oriented programming
Data structures
object oriented design and analysis
Database system
Computer networks
Discrete mathematics
Web application
I am expert in different computer languages such as C++, java, JavaScript, Sql, CSS, Python and C#. I'm also have excellent knowledge of essay writing and research. I have worked in other Freelancing website such as Fiverr and Upwork. Now I have finally decided to join the SolutionInn platform to continue with my explicit work of helping dear clients and students to achieve their academic dreams. I deliver plagiarism free work and exceptional projects on time. I am capable of working under high pressure.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Lakefield Corporations oil trading desk buys and sells oil products (crude oil and refined fuels), options, and futures in international markets. The trading desk is responsible for buying raw...
-
Read the case study "Southwest Airlines," found in Part 2 of your textbook. Review the "Guide to Case Analysis" found on pp. CA1 - CA11 of your textbook. (This guide follows the last case in the...
-
A stationary jet engine is shown. Air with a density of 0.0805 lb/ft 3 enters as shown. The inlet and outlet cross sectional areas are both 10.8 ft 2 . The mass of fuel consumed is 2% of the mass of...
-
Graph the sets of points whose polar coordinates satisfy the equations and inequalitie. 0 , r = 1
-
Go to Case 21.3,Spray-Tek, Inc. v. Robbins Motor Transportation, Inc., 426 F.Supp.2d 875 (W.D. Wis. 2006), on pages 428430. Read the excerpt and answer the following questions. (a) Issue: What...
-
Cummings Products Company is considering two mutually exclusive investments. The projects' expected net cash flows are as follows: a. Construct NPV profiles for Projects A and B. b. What is each...
-
Leicht Transfer & Storage provides warehousing services and often purchases pallets from Pallet Central. The companies followed a standard practice for documenting these transactions in which Pallet...
-
The Conch Oil Company needs to transport 30 million barrels of crude oil from a port in Doha, Qatar in the Persian Gulf to three refineries throughout Europe. The refineries are in Rotterdam,...
-
A single nuclear reactor produces 2.6GW of electrical power, and has a generator voltage of 22kV.What percentage of power would be lost from our nuclear reactor in three-line transmission that goes...
-
Ken Ham started his own consulting firm, Hambone Consulting, on May 1, 2010. The trial balance at May 31 is as follows. In addition to those accounts listed on the trial balance, the chart of...
-
What is the difference between a crystalline solid and an amorphous solid?
-
Explain the difference between a pure substance and a mixture
-
Evaluate the indefinite integral. 1 + x 1 + x dx
-
Identify the advantage of a partnership. ( Check all that apply ) . Each partner has unlimited liability for the partnership's debts. Partenership interest is not freely transferable. It is easy and...
-
To save money, the Bunny Brigade has been holding recent meetings in Mr. McGregor's garden. Like the squirrels, the bunnies use maple leaves (ML) as currency. The Bunny Brigade will charge bunnies...
-
Dains Diamond Bit Drilling purchased the following assets this year. Asset Purchase Date Original Basis Drill bits (5-year) January 25 $ 90,000 Drill bits (5-year) July 25 95,000 Commercial building...
-
How to complete profitability ratios and liquidity ratios for Macys financial analysis?
-
How can you use this evidence to examine the accuracy of inventory balance on B/S?
-
Two income statements for Hemlock Company are shown below. a. Prepare a vertical analysis of Hemlock Companys income statements. b. Does the vertical analysis indicate a favorable or an...
-
A random sample of 10 houses heated with natural gas in a particular area, is selected, and the amount of gas (in therms) used during the month of January is determined for each house. The resulting...
-
Calculate the energy in kJ necessary to vaporize 100 g of water at its boiling point.
-
Octane has a normal boiling point of 125.5 o C and a heat of vaporization (H vap ) of 38.8 kJ / mol. What is the vapor pressure of octane at 20 o C?
-
A form of iron crystallizes with a body-centered unit cell. The radius of iron is 124 pm. Calculate the density of this form of iron in g/cm 3 .
-
Housing developments in Salt Lake City. Landlords across the country are competing to keep tenants, especially at new buildings. When Tressy Coats looked to rent a somewhat worn-out, three-bedroom...
-
Set the GDP expenditure to search for the 4th Quarter data from last year (example Q4/2021). Set Prices: to "Current prices," and set Seasonal adjustment: to "Seasonally adjusted at annual rates."...
-
Cocoa price per tonne (Variable Costs): find today's spot price of cocoa per tonne. Use the following website to determine the US$/tonne of cocoa:...

Study smarter with the SolutionInn App


